Chapter: Essential Anesthesia From Science to Practice : Applied physiology and pharmacology : A brief pharmacology related to anesthesia
Pharmacologic preparation for anesthesia
Pharmacologic preparation for anesthesia
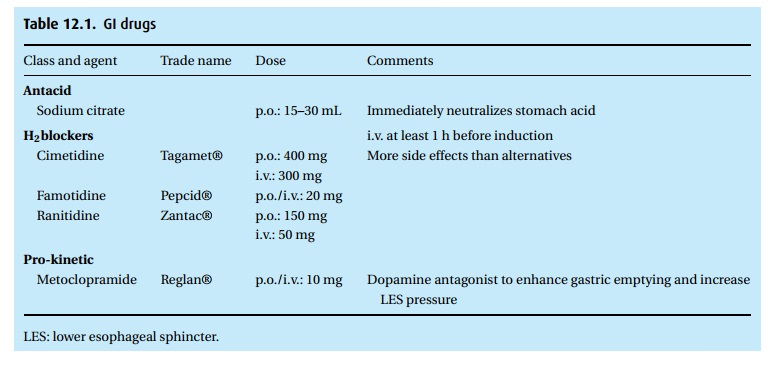
Reduce the risk of aspiration (Table 12.1)
The
aspiration of acid gastric juice can lead to a nasty chemical burn of the
trachea and bronchi and to bronchospasm and pneumonitis and, potentially, to
death. We aim to reduce gastric volume and limit acidity. Gastric juice with a
pH of 2.5 or less is thought to cause dangerous chemical burns when aspirated.
We have several methods to reduce the hazards of aspiration of acidic juice:
·
Buffer the gastric acid with an antacid. Many different agents are
available. We prefer a non-particulate liquid, which not only mixes more
readily in the stomach but also causes less harm when aspirated than would be
true for a particulate antacid. Sodium citrate (trisodium citrate) or Bicitra®
(sodium citrate and citric acid) – which are liquid – find common use in
anesthe-sia. We give 15–30 mL by mouth within 30 minutes before induction of
anesthesia.
·
Enhance gastric emptying. Metoclopramide (Reglan®) works both
locally – acetylcholine-like and thus enhancing lower esophageal sphincter
tone, gas-tric motility and emptying – and centrally as a dopaminergic blocker.
We do not know how much the CNS action contributes to the desired GI effect,
but we do know that the drug can cause undesirable CNS effects, inclu-ding
extrapyramidal symptoms; it might contribute to early post-operative delirium.
Typical doses for the average adult are 10 to 20 mg by mouth 1 hr (or 10 mg
i.v. 30 minutes) before anesthesia.
·
Inhibit gastric secretion. We have several drugs that antagonize H2
receptors and thus inhibit secretion of gastric acid, among them cimetidine
(Taga-met®), ranitidine (Zantac®), and famotidine (Pepcid®). We prefer
ranitidine (150 mg p.o. or 50 mg i.v.) or famotidine (20 mg p.o. or i.v.) an
hour before anesthesia. Proton pump inhibitors (among them omeprazole
(Prilosec®), esomeprazole (Nexium®) and pantoprazole (Protonix®)) can also
reduce gastric acidity. Because of their slow onset of action (hours), proton
pump inhibitors are not routinely prescribed as antacids in anesthesia.
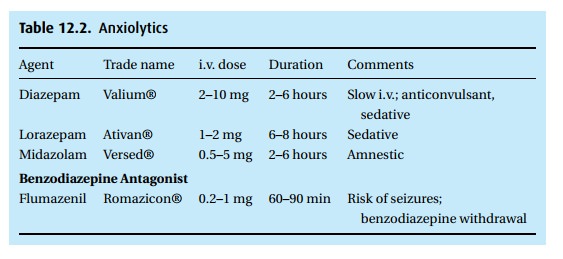
Reduce anxiety (Table 12.2)
Benzodiazepines
To allay
fear and induce antegrade amnesia, many patients receive a benzodia-zepine
before induction of anesthesia. Several different benzodiazepines are on the
market. Prominent among them is diazepam (Valium®) and midazolam (Versed®); the
latter is about three times as potent as diazepam.
In most adults, small (1–2 mg) intravenous doses of midazolam produce not only a calming effect, but also antegrade amnesia. The effect sets in over 2 to 3 minutes. Benzodiazepines work through GABA receptors, much like alcohol, and therefore, those who are not alcohol-na¨ıve might require additional doses. However, the doses should be separated by at least 2 minutes to avoid missing respiratory depression and even apnea. The elimination half-life is about 3 hours.
Midazolam
can reduce the incidence of recall of intra-operative events.
Midazolam
has also been used to induce anesthesia. We slowly administer 0.2 to 0.3 mg/kg
intravenously, and anticipate respiratory depression. Even in small doses, e.g.,
1 mg for the average adult, the drug serves as a good anticonvulsant.
As with
all CNS active drugs, we use great care in fear of drug interaction, as may
occur at the extremes of age or in the debilitated patient.
Flumazenil (Romazicon®) antagonizes the effects of benzodiazepines
(see Table 12.2). We titrate it to effect,
starting with 0.2 mg given slowly intravenously and not more than a total of 3
mg for the average adult. In case of midazolam-induced respiratory depression,
we would manually ventilate the patient’s lungs rather than start with an
antagonist. Flumazenil can trigger convulsions when given to patients poisoned
with tricyclic antidepressants or chronically on high doses of benzodiazepines.
Prevent nausea and vomiting
(Table 12.3)
Even though
modern anesthesia techniques have decreased the frequency of early
postoperative nausea and vomiting, these two disagreeable complications still
trouble patients greatly. A number of drugs help to suppress or minimize the
occurrence.
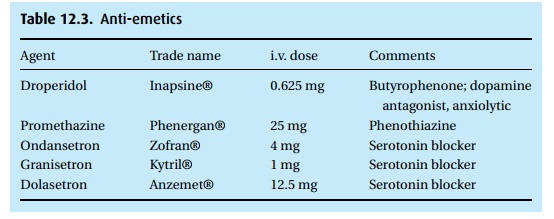
Droperidol (Inapsine®)
This butyrophenone is a dopamine antagonist. It has been around for
over 30 years and has been used extensively during anesthesia and for the
prevention or treat-ment of nausea and vomiting. We start with 0.625 mg i.v. to
the average adult. The question has been raised whether it would be justifiable
to give droperidol pro-phylactically, which would mean giving it to many
patients who would not have developed nausea and vomiting. Such across the
board prophylaxis can only be defended when the drug poses no risk but offers
considerable benefits. Droperidol
offers
the benefits but not without risks. In 2001, the FDA published a warning
implicating droperidol in the prolongation of the QT interval (normal between
0.38 s and 0.42 s, with fast or slow heart rates, respectively). It quoted
studies describing patients who developed widening QT intervals exceeding 0.45
s and ending in torsade de pointes, a malignant arrhythmia. Many drugs have
been shown to prolong the QT interval, more frequently in women than men. The
list includes (but is not limited to) amiodarone (Cordarone®), cisapride
(Propul-sid®), erythromycin, quinidine, and sotalol (Betapace®). We must be
particularly concerned in patients with existing prolongation of the QT
interval. We mention the worry about QT prolongation even though in anesthesia
a dangerous pro-longation of QT intervals had not been linked to droperidol.
However, the issue raised by the FDA has caused considerable discussion in
anesthesia circles.
Droperidol
has other side effects that may be quite troublesome, if not lethal. Very few
patients develop extrapyramidal symptoms, others a feeling of terror which they
cannot express.
Serotonin receptor blockers
Among
these are ondansetron (Zofran®), granisetron (Kytril®), and dolasetron
(Anzemet®). These serotonin receptor blockers have found use in patients
undergoing chemotherapy and in the prevention of nausea and vomiting
post-operatively. The drugs appear more useful as a prophylactic antiemetic
rather than in treatment of existing nausea and vomiting. Fortunately, these
agents are not burdened with a list of disagreeable side effects (unless you
count their cost!) – other than constipation in 11% of patients, something of
concern to patients undergoing chemotherapy, and even less frequent headaches
and elevated liver enzymes.
Related Topics