Chapter: Biotechnology Applying the Genetic Revolution: Noninfectious Diseases
The Insulin Receptor
THE
INSULIN RECEPTOR
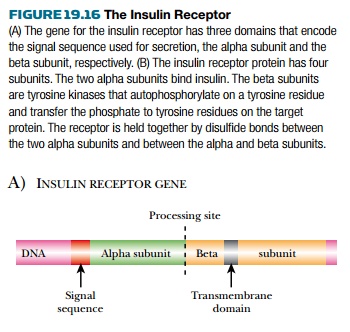
The insulin receptor is a tetramer of
two alpha and two beta chains ( Fig. 19.16 ). Just as in insulin, a single gene
produces a single protein product that is processed to release the signal sequence
and then to give the separate alpha and beta chains of the receptor. The alpha chains
are exposed on the cell surface and bind the hormone. Disulfide bonds form
between the cysteinerich regions of the two alpha chains and hold them
together. The paired beta chains are embedded in the membrane and possess an
internal signal-transmitting domain with protein kinase activity. A variety of genetic
defects may affect the insulin receptor. Lowered expression of the receptor
gene, defects in processing, defective insulin binding, and defects in protein
kinase activity are all known. The various syndromes that result are resistant
to insulin treatment.
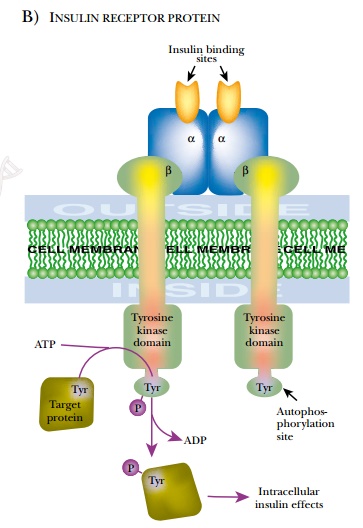
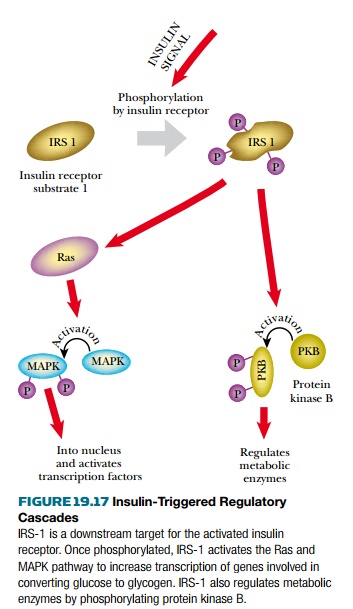
In normal insulin receptors, binding of
insulin triggers a conformational change that activates the protein kinase
domain. This initiates two alternative phosphorylation cascades. The protein
kinase B pathway is responsible for short-term control of glucose metabolism
and involves no new protein synthesis ( Fig. 19.17 ). The other pathway
proceeds via the signal transmission proteins Ras and MAPK. This leads to gene
activation and the synthesis of new proteins, over a longer time scale. The
details of insulin action vary between different types of target cells (e.g.,
liver, fat, or muscle). Overall, insulin lowers blood glucose and promotes its
storage as glycogen. If blood sugar is too low, the hormone glucagon acts in
oppositionto insulin, promoting the breakdown of glycogen and the release of
more glucose into the bloodstream. Glucagon uses cyclic AMP as second
messenger.
Related Topics