Chapter: Biotechnology Applying the Genetic Revolution: Noninfectious Diseases
Obesity and Leptin
OBESITY
AND LEPTIN
Our word obesity comes from the
Latin obedere , meaning “to devour.” Overeating and the accompanying consequences
have become epidemic in the industrialized nations. Americans spend more than $12
billion a year on slimming products (diets, exercise machines, liposuction,
etc.), yet they are the most overweight people of any industrial nation and are
getting fatter. About 35 million Americans are sufficiently overweight to
seriously affect their health. The increased health costs amount to some $40
billion per year, more than three times the amount spent on slimming products!
Obesity is not just a matter of diet and (lack of) exercise, but depends
greatly on genetics.
The ob (obese) gene encodes a
protein hormone called leptin . Mice defective in both copies of the ob gene
lack leptin and weigh up to three times as much as normal mice. When obese mice
are treated with leptin, they not only eat much less, but also burn off fat
much faster and may lose 30% of their body weight. The genes for human leptin
and mouse leptin have both been cloned into the bacterium E. coli , allowing
the hormones to be made in large quantities. Leptin from both species is very
similar and acts in the same way on obese mice.
Human trials using cloned human leptin
have shown only moderate effects. Some test subjects lost a dozen pounds over 6
months. However, leptin has little effect on most very obese people, indicating
that other factors are important. Leptin defects cause gross obesity, which
appears almost from birth and is technically known as morbid obesity .
This is a relatively rare condition in humans. Unlike obesity in mice, few humans
have been found who have defective ob genes and whose obesity is due to
lack of leptin. Another reason why leptin is unlikely to be widely used is that
digestive enzymes degrade it. So leptin cannot be taken by mouth but has to be
injected daily.
Other obese humans have defects in other
components of the leptin regulatory circuit. Many obese humans actually have higher than normal
leptin levels and may suffer from defects in the leptin receptor . Mice lacking
leptin receptor, due to mutation of the db gene , have two to three
times the normal levels of leptin in their blood. The leptin receptor is found
on the surface of brain cells, especially in the hypothalamus. It binds leptin
that is circulating in the bloodstream and so passes the signal on to the brain
( Fig. 19.21 ).
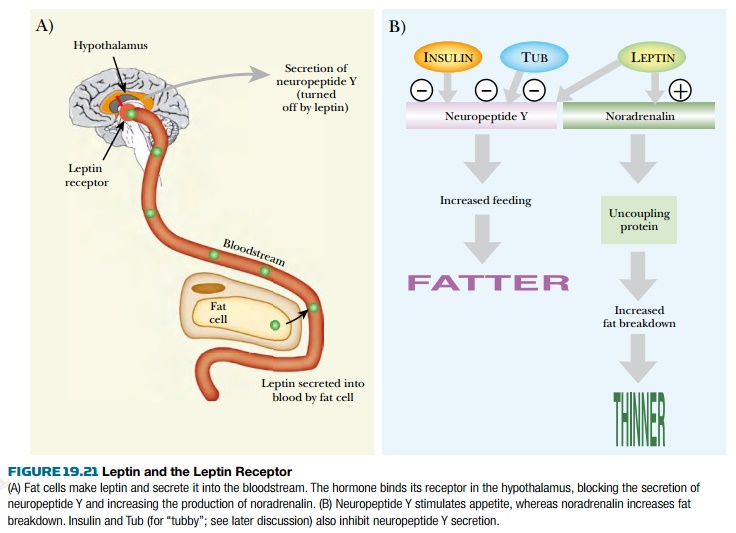
When leptin binds to its receptor, it
turns off the release of neuropeptide Y by the brain. Neuropeptide Y increases
feeding and so makes animals fatter. In addition, leptin stimulates the release
of noradrenalin (= norepinephrine ) by the nervous system. This binds to b 3-adrenergic
receptors on the surfaces of fat cells and signals them to burn off fat.
Partially defective β3-adrenergic receptors have been found in some overweight
people. The leptin receptor has a cytoplasmic tail that acts as a protein
kinase and is responsible mfor transmitting the signal. When leptin binds, it
initiates a phosphotransfer cascade that ultimately triggers release of
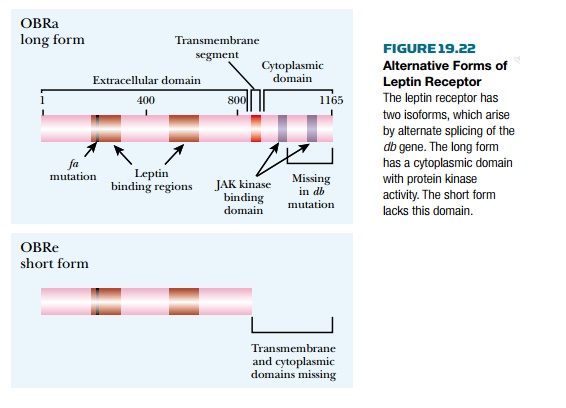
noradrenaline. However, the mRNA for the leptin receptor undergoes alternative
splicing to give proteins of different lengths ( Fig. 19.22 ). The longer isoform
is found in the hypothalamus and activates the multiple signaling pathways described
earlier. The shorter isoform lacks the cytoplasmic kinase domain and its role
is unclear, although it may bind leptin and carry it across the blood-brain
barrier. As noted earlier, db mutants lack receptor completely. However,
in fa mutants the receptor is present but
has a damaged signaling tail. (The fa mutation, found originally in
rats, is in the db gene, encoding the leptin receptor, not in the
fat gene.)
Related Topics