Chapter: Biotechnology Applying the Genetic Revolution: Noninfectious Diseases
A Multitude of Genes Affect Obesity
A
MULTITUDE OF GENES AFFECT OBESITY
Fat accumulation is affected by many
factors, both genetic and environmental. Most cases of obesity are not due to
defects in leptin or its receptor. Nonetheless, the discovery that at least some
cases of obesity are largely genetic has created a major research effort in
this area. In addition, there are the potential profits from drugs for
controlling appetite and body weight. An assortment of genes and proteins now
known to affect body weight and fat accumulation is listed in Table 19.1 .
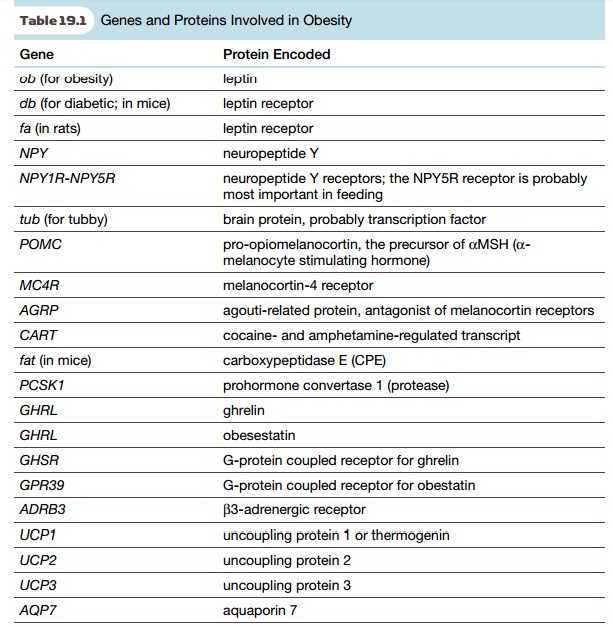
This list gives a confusing choice of
genes on which to blame obesity. It also gives a large number of possible targets
for drug development. Unfortunately, a change in any one factor tends to affect
the level of other components of the regulatory system. Consequently, the
development of a “magic bullet” to control obesity is unlikely. Exactly how the
overall control network is arranged remains for future research to uncover.
Insulin not only controls sugar levels
in the blood but also plays a role in fat metabolism. Furthermore, insulin
receptors are present on some brain cells, thus linking insulin to the control
of eating and body fat. Insulin decreases feeding by regulating neuropeptide Y.
As discussed earlier, insulin is secreted as the inactive precursor proinsulin,
which must beprocessed to insulin in order to act. Carboxypeptidase E is
anenzyme that processes proinsulin into active insulin. In mice referred to
technically as “fat,” there is a genetic defect in the gene for
carboxypeptidase E. These mice therefore accumulate proinsulin. They also resemble
many overweight people in growing fatter as they age, illustrating how lowered
insulin levels contribute to fat accumulation.
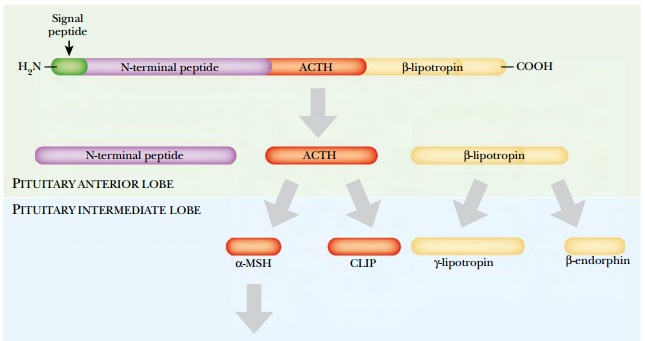
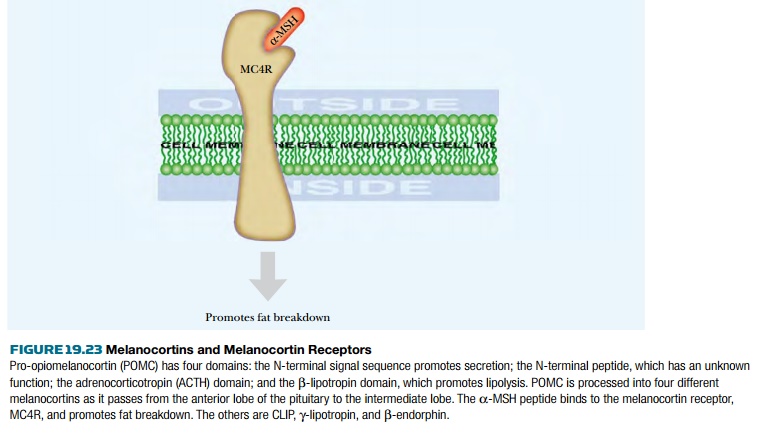
Another set of regulators that affect fat metabolism is the melanocortins ( Fig. 19.23 ). Pro-opiomelanocortin (POMC) is a precursor protein that is cleaved to give several different melanocortins. Of these, a-melanocyte stimulating hormone ( aMSH) affects fat accumulation. There are also several different melanocortin receptors . Of these, melanocortin-4 receptor (MC4R) is needed by aMSH to promote fat breakdown. Defects in the POMC or MC4R genes may result in obesity. How melanocortin control interacts with other regulatory factors is still being debated. Another defect is found in “tubby” mice, defective in the tub gene whose function is still uncertain. Defects in tub cause moderate accumulation of fat later in life. The Tub protein is present in the hypothalamus, the same area of the brain targeted by leptin. It also seems to regulate the level of neuropeptide Y, but by a mechanism different from leptin or melanocortin. Ghrelin is a recently discovered 28-amino-acid peptide that has major effects on feeding behavior. The levels of ghrelin in blood rise as mealtimes approach and fall afterward, suggesting that ghrelin plays a major role in signaling hunger. Ghrelin was originally named as a “growth hormone releasing factor” but has other effects and appears to block leptin action. Injecting rats with ghrelin stimulates them to eat more and gain weight. Obesestatin is a related peptide hormone that opposes the action of ghrelin. Fascinatingly, both ghrelin and obesestatin are derived from processing of the same prohormone, proghrelin.
Related Topics