Chapter: Basic & Clinical Pharmacology : Opioid Analgesics & Antagonists
Strong Agonists
SPECIFIC AGENTS
The following section
describes the most important and widely used opioid analgesics, along with
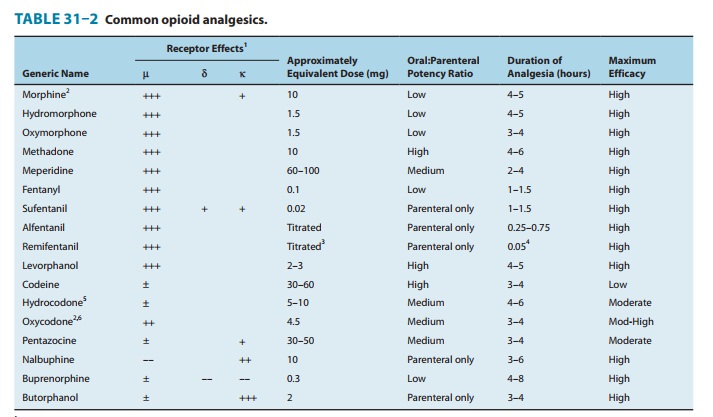
features peculiar to specific agents. Data about doses approximately equivalent
to 10 mg of intramuscular morphine, oral versus parenteral efficacy, duration
of analgesia, and intrinsic activity (maximum efficacy) are pre-sented in Table
31–2.
STRONG AGONISTS
Phenanthrenes
Morphine,
hydromorphone, and oxymorphone are strong ago-nists
useful in treating severe pain. These prototypic agents have been described in
detail above.
Heroin (diamorphine, diacetylmorphine) is potent and fast-acting, but
its use is prohibited in the USA and Canada. In recent years, there has been
considerable agitation to revive its use. However, double-blind studies have
not supported the claim that heroin is more effective than morphine in
relieving severe chronic pain, at least when given by the intramuscular route.
Phenylheptylamines
Methadone
has
undergone a dramatic revival as a potent andclinically useful analgesic. It can
be administered by the oral, intra-venous, subcutaneous, spinal, and rectal
routes. It is well absorbed from the gastrointestinal tract and its
bioavailability far exceeds that of oral morphine.
Methadone is not only
a potent μ-receptor
agonist but its race-mic mixture of D-
and L-methadone isomers can
also block both NMDA receptors and monoaminergic reuptake transporters. These
nonopioid receptor properties may help explain its ability to relieve
difficult-to-treat pain (neuropathic, cancer pain), especially when a previous
trial of morphine has failed. In this regard, when analgesic tolerance or
intolerable side effects have developed with the use of increasing doses of
morphine or hydromorphone, “opi-oid rotation” to methadone has provided
superior analgesia at 10–20% of the morphine-equivalent daily dose. In contrast
to its use in suppressing symptoms of opioid withdrawal, use of metha-done as
an analgesic typically requires administration at intervals of no more than 8
hours. However, given methadone’s highly variable pharmacokinetics and long
half-life (25–52 hours), initial admin-istration should be closely monitored to
avoid potentially harmful adverse effects, especially respiratory depression.
Because metha-done is metabolized by CYP3A4 and CYP2B6 isoforms in the liver,
inhibition of its metabolic pathway or hepatic dysfunction has also been
associated with overdose effects, including respiratory depres-sion or, more
rarely, prolonged QT-based cardiac arrhythmias.
Methadone is widely
used in the treatment of opioid abuse. Tolerance and physical dependence
develop more slowly with methadone than with morphine. The withdrawal signs and
symptoms occurring after abrupt discontinuance of methadone are milder,
although more prolonged, than those of morphine. These properties make
methadone a useful drug for detoxification and for maintenance of the chronic
relapsing heroin addict.
For detoxification of a heroin-dependent addict, low doses of methadone (5–10 mg orally) are given two or three times daily for 2 or 3 days. Upon discontinuing methadone, the addict experiences a mild but endurable withdrawal syndrome.For maintenance therapy of the opioid recidivist, tolerance to 50–100 mg/d of oral methadone may be deliberately produced; in this state, the addict experiences cross-tolerance to heroin, which prevents most of the addiction-reinforcing effects of heroin. One rationale of maintenance programs is that blocking the reinforcement obtained from abuse of illicit opioids removes the drive to obtain them, thereby reducing criminal activity and making the addict more amenable to psychiatric and rehabilitative therapy. The pharmaco-logic basis for the use of methadone in maintenance programs is sound and the sociologic basis is rational, but some methadone pro-grams fail because nonpharmacologic management is inadequate.
The concurrent
administration of methadone to heroin addicts known to be recidivists has been
questioned because of the increased risk of overdose death secondary to
respiratory arrest. Not only has the number of patients prescribed methadone
for persistent pain increased, but the incidence of accidental overdose and
complications related to respiratory depression have also increased.
Buprenorphine, a partial μ-receptor agonist with long-acting properties,
has been found to be effective in opioid detoxi-fication and maintenance
programs and is presumably associated with a lower risk of such overdose
fatalities.
Phenylpiperidines
Fentanyl is one of the most widely used agents in the family ofsynthetic
opioids. The fentanyl subgroup now includes sufenta-nil, alfentanil, and
remifentanil in addition to the parent com-pound, fentanyl.
These opioids differ
mainly in their potency and biodisposi-tion. Sufentanil is five to seven times
more potent than fentanyl. Alfentanil is considerably less potent than
fentanyl, but acts more rapidly and has a markedly shorter duration of action.
Remifentanil is metabolized very rapidly by blood and nonspecific tissue
esterases, making its pharmacokinetic and pharmacodynamic half-lives extremely
short. Such properties are useful when these com-pounds are used in anesthesia
practice. Although fentanyl is now the predominant analgesic in the
phenylpiperidine class, meperi-dine continues
to be used. This older opioid has significant anti-muscarinic effects, which
may be a contraindication if tachycardia would be a problem. Meperidine is also
reported to have a nega-tive inotropic action on the heart. In addition, it has
the potential for producing seizures secondary to accumulation of its
metabo-lite, normeperidine, in patients receiving high doses or with
con-current renal failure. Given this undesirable profile, use of meperidine as
a first-line analgesic is becoming increasingly rare.
Morphinans
Levorphanol
is a
synthetic opioid analgesic closely resemblingmorphine in its action.
Related Topics