Chapter: Biotechnology Applying the Genetic Revolution: Recombinant DNA Technology
Restriction Enzymes Cut DNA; Ligase Joins DNA
RESTRICTION
ENZYMES CUT DNA; LIGASE JOINS DNA
The ability to isolate,
separate, and visualize DNA fragments would be useless unless some method was
available to cut the DNA into fragments of different sizes. In fact, naturally
occurring restriction enzymes or restriction endonucleases are the key
to making DNA fragments. These bacterial enzymes bind to specific recognition
sites on DNA and cut the backbone of both strands. They evolved to protect
bacteria from foreign DNA, such as from viral invaders. The enzymes do not cut
their own cell’s DNA because they are methylation sensitive, that is, if the
recognition sequence is methylated, then the restriction enzyme cannot bind.
Bacteria produce modification enzymes
that recognize the same sequence as the corresponding restriction enzyme. These
methylate each recognition site in the bacterial genome. Therefore, the
bacteria can make the restriction enzyme without endangering their own DNA.
Restriction enzymes have been
exploited to cut DNA at specific sites, since each restriction enzyme has a
particular recognition sequence. Differences in cleavage site determine the
type of restriction enzyme. Type I restriction enzymes cut the DNA strand 1000
or more base pairs from the recognition sequence. Type II restriction enzymes
cut in the middle of the recognition sequence and are the most useful for
genetic engineering. Type II restriction enzymes can either cut both strands of
the double helix at the same point, leaving blunt ends, or they can cut at
different sites on each strand leaving single- stranded ends, sometimes called
sticky ends (Fig. 3.2). The recognition sequences of Type II restriction
enzymes are usually inverted repeats, so that the enzyme cuts between the same
bases on both strands. Since the repeats are inverted, the cuts may be
staggered, thus generating single-stranded overhangs. Some commonly used
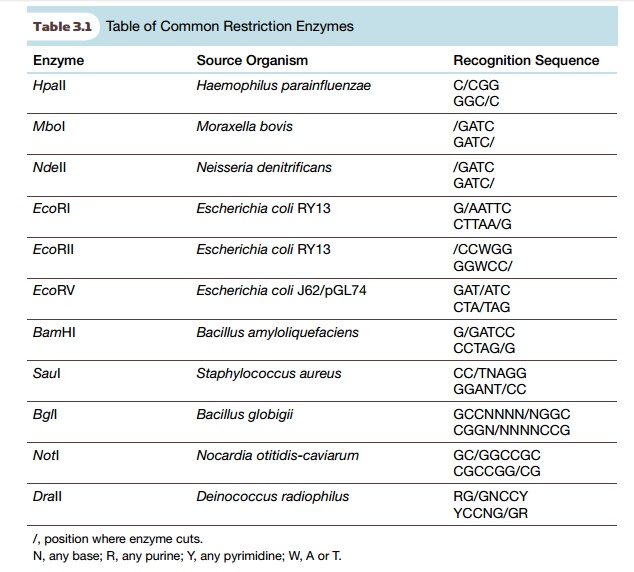
restriction enzymes for biotechnology applications are listed in Table 3.1.
The number of base pairs in
the recognition sequence determines the likelihood of cutting. Finding a
particular sequence of four nucleotides is much more likely than finding a six
base-pair recognition sequence.
So to generate fewer, longer
fragments, restriction enzymes with six or more base-pair recognition sequences
are used. Conversely, four base-pair enzymes give more, shorter fragments from
the same original segment of DNA.
When two different DNA
samples are cut with the same sticky-end restriction enzyme, all the fragments
will have identical overhangs. This allows DNA fragments from two sources
(e.g., two different organisms) to be linked together (Fig. 3.3). Fragments are
linked or ligated using DNA ligase, the same enzyme that ligates the Okazaki
fragments during replication. The most common ligase used is actually from T4
bacteriophage. Ligase catalyzes linkage between the 3′-OH of one strand and the
5′-PO4 of the other DNA strand. Ligase
is much more efficient with overhanging sticky ends, but can also link blunt
ends much more slowly. (Specific fragments for ligation are often isolated by
agarose gel electrophoresis as described earlier.)

Related Topics