Chapter: Biotechnology Applying the Genetic Revolution: DNA Synthesis in Vivo and in Vitro
Polymerase Chain Reaction Uses In Vitro Synthesis to Amplify Small Amounts of DNA
POLYMERASE
CHAIN REACTION USES IN VITRO
SYNTHESIS TO AMPLIFY SMALL AMOUNTS OF DNA
The polymerase chain reaction (PCR) amplifies small samples of DNA into
large amounts, much as a photocopier makes many copies of one sheet of paper.
The DNA is amplified using the principles of replication, that is, the DNA is
replicated over and over by DNA polymerase until a large amount is
manufactured. Kary Mullis invented this technique while working at Cetus in
1987. He eventually won the Nobel Prize in Chemistry for PCR because of its
huge impact on biology and science. PCR is used in forensic medicine to identify
victims or criminals by amplifying the minuscule amounts of DNA left at a crime
scene; PCR can identify infectious diseases such as AIDS before symptoms
emerge; PCR can amplify specific segments of genes without the need for cloning
the segment first; in fact, PCR is now used in all aspects of the biological
sciences.
Just as the photocopier needs more paper, ink, and a mechanism to copy, PCR requires specific reagents. The sample to be copied is called the template DNA, and this is often a known sequence or gene. The template DNA is double-stranded, and extremely small quantities are sufficient. PCR can amplify a particular gene segment directly from one Caenorhabditis elegans or even a single cell. The second reagent needed for PCR is a pair of primers, which have sequences complementary to the ends of the template DNA. The DNA primers are oligonucleotides about eight to 20 nucleotides long. One primer anneals to the 5′ end of the sense strand, and the other anneals to the 3′ end of the antisense strand of the target sequence. Because the primers are specific, the template can be mixed with other DNA sequences. The third reagent is a supply of nucleoside triphosphates, and the fourth is Taq DNA polymerase from Thermus aquaticus, which actually makes the copies.
The basic mechanism of PCR relies on heat denaturation of the template, annealing of the primers, and making a complementary copy using DNA polymerase, all being steps found in DNA replication. The three steps are repeated over and over until one template strand generates millions of identical copies. An amount of DNA too small to be seen can be exponentially copied so that it can be cloned into a vector, or visualized on an agarose gel. The process requires changing the temperature in a cyclic manner. Changing temperatures is accomplished by a thermocycler, a machine designed to change the temperature of its heat block rapidly so that each cycle can be completed in minutes. The temperature cycles between 94°C to denature the template; 50–60°C to anneal the primer (depending on the length and sequence of the primer); and 70°C for Taq polymerase to make new DNA. Before thermocyclers were developed, PCR was accomplished by moving the mixture among three different water baths at different temperatures every few minutes, which was very tedious.
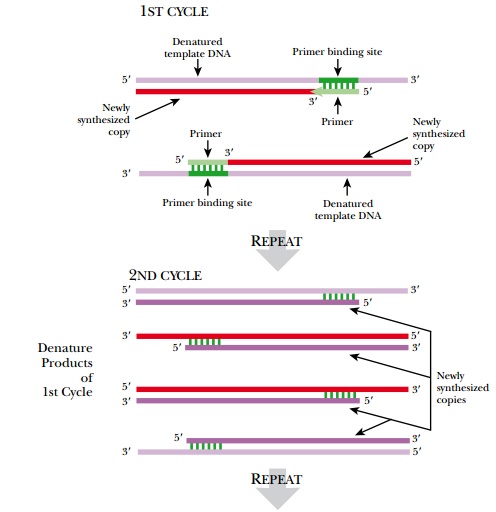
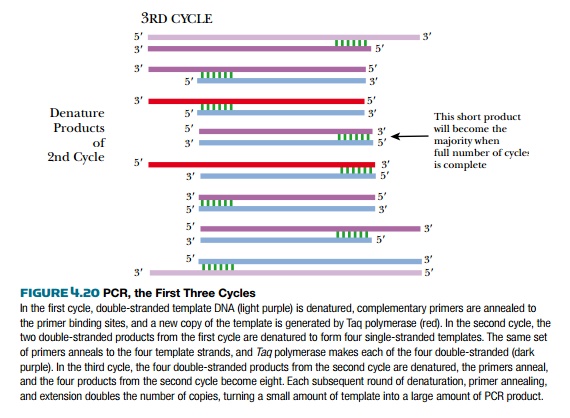
In principle, the PCR cycle resembles DNA replication with a few modifications (Fig. 4.20). Like other in vitro DNA synthesis reactions, the double-stranded template is denatured with high heat rather than enzymes. Then the temperature is lowered so that the primers anneal to their binding sites. The primers are made so that each binds to opposite strands of the template, one at the beginning and one at the end of the gene. Then DNA polymerase elongates both primers and converts both single template strands to double-stranded DNA. (Note: During sequencing, only one primer is used and only one strand of the template is replicated, but during PCR both strands are copied.) Taq polymerase is the most widely used polymerase for PCR because it is very stable at high temperatures and does not denature at the high temperatures needed to separate the strands of the template DNA. Taq polymerase comes from Thermus aquaticus, a bacterium that grows in the thermal vents in the ocean. After the first replication cycle, the whole process is repeated. The two DNA strands are denatured at high heat, and then the temperature drops to allow the primers to anneal to their target sequences. Taq polymerase synthesizes the next four strands, and now there are four double-stranded copies of the target sequence. Early in the process, some longer strands are generated; however, eventually only the segment flanked by the two primers is amplified. Ultimately the template strands and early PCR products become the minority. The shorter products become the majority.
The primers are key to the process of PCR. If the primers do not anneal in the correct location, if the span between the primers is too large, or if the primers form hairpin regions rather than annealing to the target, then Taq polymerase will not be able to amplify the segment. Also, if both primers anneal to the same strand, the reaction will not work. If the template has a known sequence, primers are synthesized based on the sequences upstream and downstream of the region to be amplified. Modifications exist that allow researchers to analyze unknown sequences by PCR (see later discussion).
Related Topics