Chapter: Medical Physiology: The Microcirculation and the Lymphatic System: Capillary Fluid Exchange, Interstitial Fluid, and Lymph Flow
Plasma Colloid Osmotic Pressure
Plasma Colloid Osmotic Pressure
Proteins in the Plasma Cause Colloid Osmotic Pressure. In the basic discussion of osmotic pressure, it was pointed out that only those molecules or ions that fail to pass through the pores of a semiperme-able membrane exert osmotic pressure. Because the proteins are the only dissolved constituents in the plasma and interstitial fluids that do not readily pass through the capillary pores, it is the proteins of the plasma and interstitial fluids that are responsible for the osmotic pressures on the two sides of the capillary membrane. To distinguish this osmotic pressure from that which occurs at the cell membrane, it is called either colloid osmotic pressure oroncotic pressure. The term “colloid” osmotic pressure is derived from the fact that a protein solution resembles a colloidal solu-tion despite the fact that it is actually a true molecu-lar solution.
Normal Values for Plasma Colloid Osmotic Pressure. Thecolloid osmotic pressure of normal human plasma averages about 28 mm Hg; 19 mm of this is caused by molecular effects of the dissolved protein and 9 mm by the Donnan effect—that is, extra osmotic pressure caused by sodium, potassium, and the other cations held in the plasma by the proteins
Effect of the Different Plasma Proteins on Colloid Osmotic Pressure. The plasma proteins are a mixture that con-tains albumin, with an average molecular weight of 69,000; globulins, 140,000; and fibrinogen, 400,000. Thus, 1 gram of globulin contains only half as many molecules as 1 gram of albumin, and 1 gram of fibrinogen contains only one sixth as many molecules as 1 gram of albumin. It should be recalled from the discussion of osmotic pressure that osmotic pressure is determined by the number of molecules dissolved in a fluid rather than by the mass of these molecules. Therefore, when corrected for number of molecules rather than mass, the following chart gives both the relative mass concentrations (g/dl) of the different types of proteins in normal plasma and their respective contributions to the total plasma colloid osmotic pressure (Πp).
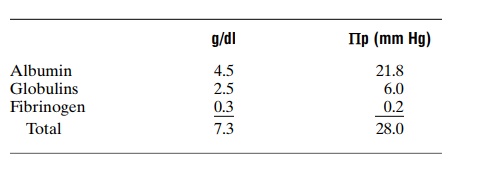
Thus, about 80 per cent of the total colloid osmotic pressure of the plasma results from the albumin frac-tion, 20 per cent from the globulins, and almost none from the fibrinogen. Therefore, from the point of view of capillary and tissue fluid dynamics, it is mainly albumin that is important.
Related Topics