Chapter: Medical Physiology: The Microcirculation and the Lymphatic System: Capillary Fluid Exchange, Interstitial Fluid, and Lymph Flow
Lymphatic System
Lymphatic System
The lymphatic system represents an accessory route through which fluid can flow from the interstitial spaces into the blood. Most important, the lymphatics can carry proteins and large particulate matter away from the tissue spaces, neither of which can be removed by absorption directly into the blood capil-laries. This return of proteins to the blood from the interstitial spaces is an essential function without which we would die within about 24 hours.
Lymph Channels of the Body
Almost all tissues of the body have special lymph chan-nels that drain excess fluid directly from the interstitial spaces. The exceptions include the superficial portions of the skin, the central nervous system, the endomysium of muscles, and the bones. But, even these tissues have minute interstitial channels called prelymphatics through which interstitial fluid can flow; this fluid even-tually empties either into lymphatic vessels or, in the case of the brain, into the cerebrospinal fluid and then directly back into the blood.
Essentially all the lymph vessels from the lower part of the body eventually empty into the thoracic duct, which in turn empties into the blood venous system at the juncture of the left internal jugular vein and left sub-clavian vein, as shown in Figure 16–8.
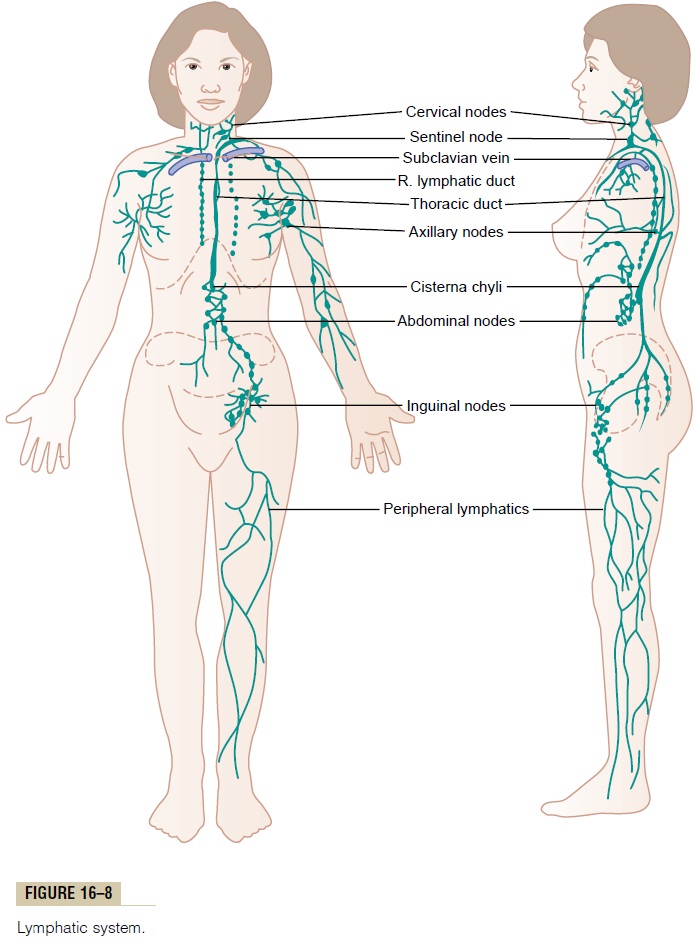
Lymph from the left side of the head, the left arm, and parts of the chest region also enters the thoracic duct before it empties into the veins.
Lymph from the right side of the neck and head, the right arm, and parts of the right thorax enters the rightlymph duct (much smaller than the thoracic duct), whichempties into the blood venous system at the juncture of the right subclavian vein and internal jugular vein.
Terminal Lymphatic Capillaries and Their Permeability. Mostof the fluid filtering from the arterial ends of blood capillaries flows among the cells andfinally is reab-sorbed back into the venous ends of the blood capil-laries; but on the average, about 1/10 of thefluidinstead enters the lymphatic capillaries and returns to the blood through the lymphatic system rather than through the venous capillaries. The total quantity of all this lymph is normally only 2 to 3 liters each day.
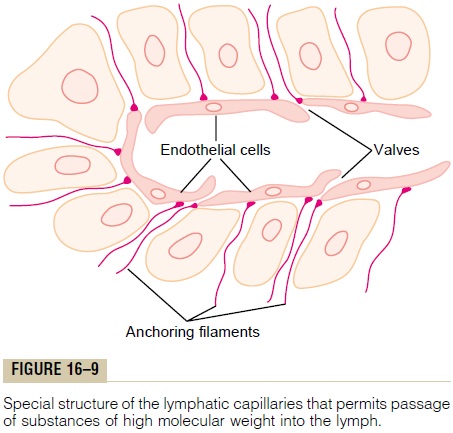
The fluid that returns to the circulation by way of the lymphatics is extremely important because sub-stances of high molecular weight, such as proteins, cannot be absorbed from the tissues in any other way, although they can enter the lymphatic capillaries almost unimpeded. The reason for this is a special structure of the lymphatic capillaries, demonstrated in Figure 16–9. This figure shows the endothelial cells of the lymphatic capillary attached by anchoring fila-ments to the surrounding connective tissue. At thejunctions of adjacent endothelial cells, the edge of one endothelial cell overlaps the edge of the adjacent cell in such a way that the overlapping edge is free to flap inward, thus forming a minute valve that opens to the interior of the lymphatic capillary. Interstitial fluid, along with its suspended particles, can push the valve open and flow directly into the lymphatic capillary. But this fluid has difficulty leaving the capillary once it has entered because any backflow closes the flap valve. Thus, the lymphatics have valves at the very tips of the terminal lymphatic capillaries as well as valves along their larger vessels up to the point where they empty into the blood circulation.
Formation of Lymph
Lymph is derived from interstitial fluid that flows into the lymphatics. Therefore, lymph as it first enters the terminal lymphatics has almost the same composition as the interstitial fluid.
The protein concentration in the interstitial fluid of most tissues averages about 2 g/dl, and the protein con-centration of lymph flowing from these tissues is near this value. Conversely, lymph formed in the liver has a protein concentration as high as 6 g/dl, and lymph formed in the intestines has a protein concentration as high as 3 to 4 g/dl. Because about two thirds of all lymph normally is derived from the liver and intes-tines, the thoracic duct lymph, which is a mixture of lymph from all areas of the body, usually has a protein concentration of 3 to 5 g/dl.
The lymphatic system is also one of the major routes for absorption of nutrients from the gastrointestinal tract, especially for absorption of virtually all fats in food. Indeed, after a fatty meal, thoracic duct lymph sometimes contains as much as 1 to 2 per cent fat.
Finally, even large particles, such as bacteria, can push their way between the endothelial cells of the lymphatic capillaries and in this way enter the lymph.
As the lymph passes through the lymph nodes, these particles are almost entirely removed and destroyed.
Rate of Lymph Flow
About 100 milliliters per hour of lymph flows through the thoracic duct of a resting human, and approxi-mately another 20 milliliters flows into the circulation each hour through other channels, making a total esti-mated lymph flow of about 120 ml/hr or 2 to 3 liters per day.
Effect of Interstitial Fluid Pressure on Lymph Flow. Figure16–10 shows the effect of different levels of interstitial fluid pressure on lymph flow as measured in dog legs.
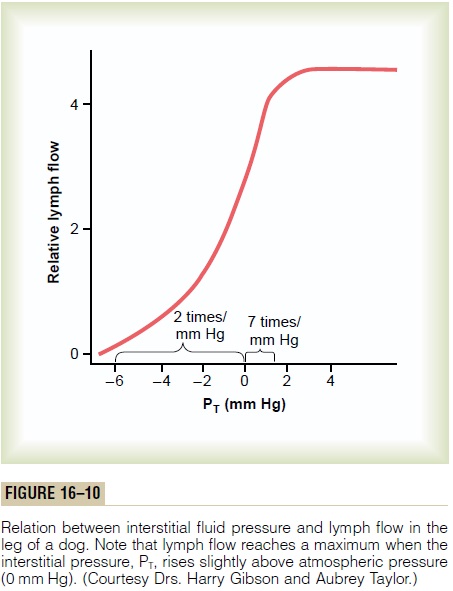
Note that normal lymph flow is very little at intersti-tial fluid pressures more negative than the normal value of -6 mm Hg. Then, as the pressure rises to 0 mm Hg (atmospheric pressure), flow increases more than 20-fold. Therefore, any factor that increases interstitial fluid pressure also increases lymph flow if the lymph vessels are functioning normally. Such factors include the following:
· Elevated capillary pressure
· Decreased plasma colloid osmotic pressure
· Increased interstitial fluid colloid osmotic pressure
· Increased permeability of the capillaries
All of these cause a balance of fluid exchange at the blood capillary membrane to favor fluid movement into the interstitium, thus increasing interstitial fluid volume, interstitial fluid pressure, and lymph flow all at the same time.
However, note in Figure 16–10 that when the inter-stitial fluid pressure becomes 1 or 2 millimeters greater than atmospheric pressure (greater than 0 mm Hg), lymph flow fails to rise any further at still higher pres-sures. This results from the fact that the increasing tissue pressure not only increases entry of fluid into the lymphatic capillaries but also compresses the outside surfaces of the larger lymphatics, thus imped-ing lymph flow. At the higher pressures, these two factors balance each other almost exactly, so that lymph flow reaches what is called the “maximum lymph flow rate.” This is illustrated by the upper level plateau in Figure 16–10.
Lymphatic Pump Increases Lymph Flow. Valves exist in alllymph channels; typical valves are shown in Figure 16–11 in collecting lymphatics into which the lym-phatic capillaries empty.

Motion pictures of exposed lymph vessels, both in animals and in human beings, show that when a collecting lymphatic or larger lymph vessel becomes stretched with fluid, the smooth muscle in the wall of the vessel automatically contracts. Furthermore, each segment of the lymph vessel between successive valves functions as a separate automatic pump. That is, even slight filling of a segment causes it to contract, and the fluid is pumped through the next valve into the next lymphatic segment. This fills the subsequent segment, and a few seconds later it, too, contracts, the process continuing all along the lymph vessel until the fluid is finally emptied into the blood circulation. In a very large lymph vessel such as the thoracic duct, this lym-phatic pump can generate pressures as great as 50 to 100 mm Hg.
Pumping Caused by External Intermittent Compression of the Lymphatics. In addition to the pumping causedby intrinsic intermittent contraction of the lymph vessel walls, any external factor that intermittently compresses the lymph vessel also can cause pumping. In order of their importance, such factors are:
· Contraction of surrounding skeletal muscles
· Movement of the parts of the body
· Pulsations of arteries adjacent to the lymphatics
· Compression of the tissues by objects outside the body
The lymphatic pump becomes very active during exer-cise, often increasing lymph flow 10- to 30-fold. Con-versely, during periods of rest, lymph flow is sluggish, almost zero.
Lymphatic Capillary Pump. The terminal lymphatic capillary is also capable of pumping lymph, in addition to the lymph pumping by the larger lymph vessels. The walls of the lymphatic capillaries are tightly adherent to the sur-rounding tissue cells by means of their anchoring filaments. Therefore, each time excess fluid enters the tissue and causes the tissue to swell, the anchoring filaments pull on the wall of the lymphatic capillary, and fluid flows into the terminal lymphatic capillary through the junctions between the endothelial cells. Then, when the tissue is compressed, the pressure inside the capillary increases and causes the overlap-ping edges of the endothelial cells to close like valves. Therefore, the pressure pushes the lymph forward into the collecting lymphatic instead of backward through the cell junctions.
The lymphatic capillary endothelial cells also contain a few contractile actomyosin filaments. In some animal tissues (e.g., the bat’s wing) these fila-ments have been observed to cause rhythmical con-traction of the lymphatic capillaries in the same way that many of the small blood and larger lymphatic vessels also contract rhythmically. Therefore, it is probable that at least part of lymph pumping results from lymph capillary endothelial cell contraction in addition to contraction of the larger muscular lymphatics.
Summary of Factors That Determine Lymph Flow. From theabove discussion, one can see that the two primary factors that determine lymph flow are (1) the intersti-tial fluid pressure and (2) the activity of the lymphatic pump. Therefore, one can state that, roughly, the rateof lymph flow is determined by the product of intersti-tial fluid pressure times the activity of the lymphatic pump.
Role of the Lymphatic System in Controlling Interstitial Fluid Protein Concentration, Interstitial Fluid Volume, and Interstitial Fluid Pressure
It is already clear that the lymphatic system functions as an “overflow mechanism” to return to the circula-tion excess proteins and excess fluid volume from the tissue spaces. Therefore, the lymphatic system also plays a central role in controlling (1) the concentration of proteins in the interstitial fluids, (2) the volume of interstitial fluid, and (3) the interstitial fluid pressure. Let us explain how these factors interact.
First, remember that small amounts of proteins leak continuously out of the blood capillaries into the interstitium. Only minute amounts, if any, of the leaked proteins return to the circulation by way of the venous ends of the blood capillaries. Therefore, these proteins tend to accumulate in the interstitial fluid, and this in turn increases the colloid osmotic pressure of the interstitial fluids.
Second, the increasing colloid osmotic pressure in the interstitial fluid shifts the balance of forces at the blood capillary membranes in favor of fluid filtration into the interstitium. Therefore, in effect, fluid is translocated osmotically outward through the capil-lary wall by the proteins and into the interstitium, thus increasing both interstitial fluid volume and interstitial fluid pressure.
Third, the increasing interstitial fluid pressure greatly increases the rate of lymph flow, as explained previously. This in turn carries away the excess inter-stitial fluid volume and excess protein that has accu-mulated in the spaces.
Thus, once the interstitial fluid protein concentra-tion reaches a certain level and causes a comparable increase in interstitial fluid volume and interstitial fluid pressure, the return of protein and fluid by way of the lymphatic system becomes great enough to balance exactly the rate of leakage of these into the interstitium from the blood capillaries. Therefore, the quantitative values of all these factors reach a steady state; they will remain balanced at these steady state levels until something changes the rate of leakage of proteins and fluid from the blood capillaries.
Significance of Negative Interstitial Fluid Pressure as a Means for Holding the Body Tissues Together
Traditionally, it has been assumed that the different tissues of the body are held together entirely by connective tissue fibers. However, at many places in the body, connective tissue fibers are very weak or even absent. This occurs particularly at points where tissues slide over one another, such as the skin sliding over the back of the hand or over the face. Yet even at these places, the tissues are held together by the negative interstitial fluid pressure, which is actually a partial vacuum. When the tissues lose their negative pressure, fluid accumulates in the spaces and the condition known as edema occurs.
Related Topics