Chapter: Medical Electronics : Bio-Chemical and Non Electrical Parameter Measurement
PH Measurement
PH MEASUREMENT
The
chemical balance in the body can be determined by the ph value of blood and
other body fluids.ph is defined as the hydrogen ion concentration of a fluid.
It is the logarithm of the reciprocal value of h+ concentration. The ph
equation is given as,
Ph= - log10
[H+] = log10 1/[H+ ]
pH is the
measure of acid- base balance in a fluid, A neutral solution has the ph value
as 7. Solutions with pH value less than 7 are acidic and above 7 are basic.
Most of the body fluids are slightly basic in nature.
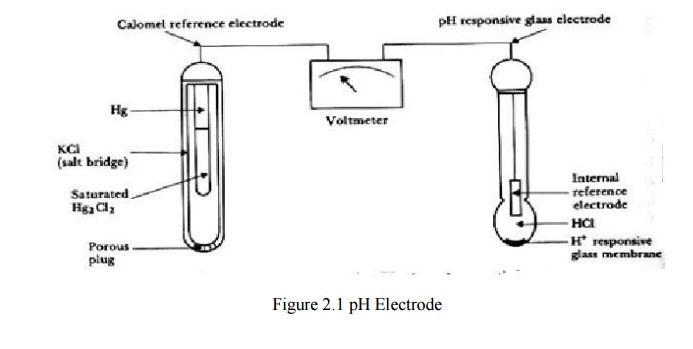
Construction and working
The ph
meter is made up of a thin glass membrane and it allows only the hydrogen ions
to pass through it. The glass electrode provides a membrane interface for H+
ions. The glass bulb at the lower end of the ph meter contains a highly acidic
buffer solution. The glass tube consists of a sliver-sliver chloride (Ag/Agcl)
electrode and the reference electrode which is made up of calomel sliver-sliver
chloride(Ag/Agcl) is tan placed in the solution in which ph is being measured.
The
potential is measured across the two electrodes. The electrochemical
measurement, which should be obtained by each of the electrodes called half-
cell. The electrode potential is called as half-cell potential. Here the glass
electrode inside the tube constitutes one half –cell and the calomel or
reference electrode is considered as the other half-cell.
For
easier ph measurement combination electrodes are used. In this type both the
active glass electrode and reference electrode are present in the same meter.
The glass electrodes are suitable only to measure ph values around 7. Since
this type of glass electrodes produce considerable errors during the
measurement of high Ph values, special type of Ph electrodes are used. After
every measurement the pH meter is washed with 20% ammonium biflouride solution,
for accurate results. The Ph meter with hydroscopic glass absorbs water readily
and provides best pH value.
Related Topics