Chapter: Genetics and Molecular Biology: Advanced Genetic Engineering
Mutagenesis with Chemically Synthesized DNA
Mutagenesis with Chemically Synthesized DNA
Khorana laid the groundwork for chemical synthesis
of DNA. He devel-oped techniques to form the phosphodiester bond between
nucleotides
Figure
10.23 Outline of one method for the
chemical synthesis of DNA. Notethat elongation proceeds from the 3’ to the 5’
end in this method.
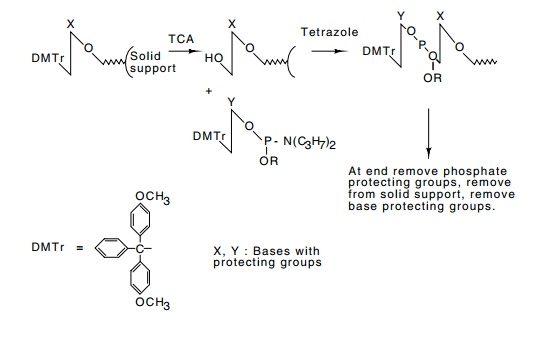
while at the same time preventing the reactive
amino, hydroxyl, and other phosphorus groups from reacting. With these
techniques, he and his co-workers then synthesized a complete tRNA gene.
Originally many person-years were required for the synthesis of 80 nucleotide
oligomers. Now, as a result of continued development by many research groups,
oligonucleotide synthesis has been highly automated and as many as 100
nucleotides may be joined in specific sequence in a day.
In chemical DNA synthesis, blocking groups are
placed on the reac - tive groups that are not to participate in the
condensation to form a phosphodiester. These are then condensed to build the
oligonucleotide (Fig. 10.23). After synthesis of the complete oligonucleotide,
all the blocking groups are removed. If the desired oligonucleotide is
particu-larly long, blocks of short, overlapping oligonucleotides can be
synthe-sized, hybridized, ligated, and finally cloned.
Before 1965, no researchers would have had a good
idea of what to do with the sequence of an entire chromosome if it were
presented to them. We are hardly in that situation now. Similarly, before 1975
there seemed to be little reason to try to synthesize DNA chemically. Not only
were relatively few interesting sequences known, but the fraction of the
synthesized material that would possess the desired sequence was likely to be
too small to be of use. With the development of cloning since 1975 and the
overall increase in our knowledge of biological mechanisms, the picture
dramatically changed. Now it is routine to synthesize a gene denovo. Convenient restriction sites can
be placed through the gene and
Figure
10.24 Mutagenesis with chemically
synthesized DNA. The oligomerhybridizes, except for the mispaired base.
Extension of the primer with DNA Pol I and ligation yields heteroduplex
molecules that can be transformed. Following DNA replication in the
transformants, the two types of DNA mole-cules segregate to yield wild-type or
mutant homoduplexes. Retransformation yields colonies containing entirely
mutant or wild-type DNA, which can be identified by hybridization with
radioactive mutant oligomer.
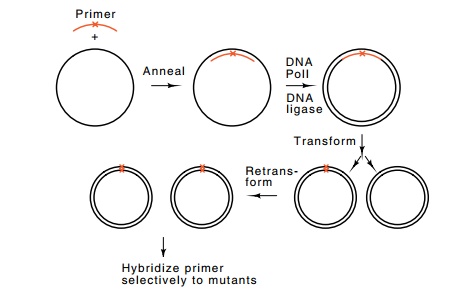
Another method for mutating a gene is to direct
mutations to a specific point. This can be done with chemically synthesized
oligonu-cleotides in a process called oligonucleotide directed mutagenesis. An
oligonucleotide containing the desired alteration, a mutation, insertion, or
deletion, will hybridize to complementary, wild-type single-stranded DNA and
can serve as a primer for DNA pol I (Fig. 10.24). The resulting double-stranded
DNA contains one wild-type strand and one mutant strand. Upon replication in
cells, one of the daughter duplexes is wild-type and the other is mutant.
Sometimes it is necessary to prevent heteroduplex repair of the mutant strand.
Either way, following trans - formation and segregation, a mutant gene can be
obtained.
Related Topics