Chapter: Obstetrics and Gynecology: Vulvar and Vaginal Disease and Neoplasia
Lichen Sclerosus - Benign Vulvar Disease
Lichen Sclerosus
Lichen
sclerosus has confused clinicians and pathologistsbecause of
inconsistent terminology and its frequent asso-ciation with other types of
vulvar pathology, including those of the acanthotic variety. As with the other
disorders, chronic vulvar pruritus occurs in most patients. Typically,the vulva is diffusely involved,
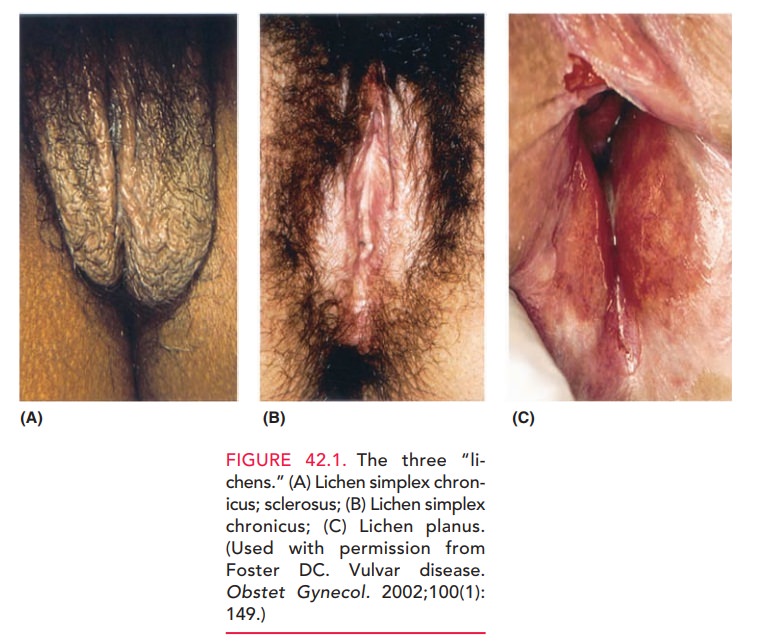
with very thin, whitish epithe-lial areas, termed “onion skin” epithelium (Fig.
42.1B). Theepithelium has been termed “cigarette paper” skin and described as
“parchment-like.” Most patients have involve-ment on both sides of the vulva,
with the most common sites being the labia majora, labia minora, the clitoral
and periclitoral epithelium, and the perineal body. The lesion may extend to
include a perianal “halo” of atrophic, whitish epithelium, forming a figure-8
configuration with the vul-var changes. In
severe cases, many normal anatomic landmarksare lost, including obliteration of
the labial and periclitoral archi-tecture as well as severe stenosis of the
vaginal introitus. Somepatients have areas of cracked skin, which are prone
to bleeding with minimal trauma. Patients with these severe anatomic changes
complain of difficulty in having normal coital function.
The etiology of lichen sclerosus is unknown, but a familial association has been noted, as well as disorders of the immune system, including thyroid disorders and Class II human leukocyte antigens.
However,
the response to topical steroids furtherindicates the underlying inflammatory
process and the role of prostaglandins and leukotrienes in the hallmark symptom
of pruritus. Histologic evaluation and
confirmation of lichen scle-rosis is often necessary and useful, because they
allow specific ther-apy. The histologic features of the lichenoid pattern
includea band of chronic inflammatory cells, consisting mostly of lymphocytes,
in the upper dermis with a zone of homoge-neous, pink-staining, collagenous-like
material beneath the epidermis due to cell death. The obliteration of
boundaries between collagen bundles gives the dermis a “hyalinized” or “glassy”
appearance. This dermal homogenization/ sclerosis pattern is virtually
pathognomonic.
In 27% to 35% of patients, there
are associated areas of acanthosis characterized by hyperkeratosis—an increase in the number of epithelial cells
(keratinocytes) with flat-tening of the rete pegs. These areas may be mixed
through-
In patients with this mixed pattern, both components need to
be treated to effect resolution of symptoms. Patients in whom a large
acanthotic component has been histologically confirmed should be treated
initially with well-penetrating cortico-steroid creams. With improvement of
these areas (usually 2 to 3 weeks), therapy can then be directed to the
lichenoid component.
Treatment
for lichen sclerosis includes the use of topical steroid (clobetasol)
preparations in an effort to ameliorate symp-toms. The
lesion is unlikely to resolve totally. Intermittenttreatment may be needed
indefinitely, which is in marked contrast to acanthotic lesions, which usually
totally resolve within 6 months.
Lichen
sclerosus does not significantly increase the patient’s risk of developing
cancer.
It has been estimated that this
risk is in the 4% range. However, due to the frequent coexistence with
acanthosis, the condition needs to be followed carefully and a rebiopsy
performed, because therapeutically resistant acanthosis can be a harbinger of
squamous cell carcinoma (SCC).
Related Topics