Chapter: Microbiology and Immunology: Antigen-Antibody Reactions
Immunofluorescence - Antigen Antibody Reactions
Immunofluorescence
The property of certain dyes absorbing light rays at one
parti-cular wavelength (ultraviolet light) and emitting them at a different
wavelength (visible light) is known as fluorescence. Fluorescent dyes, such
as fluorescein isothiocyanate and lissa-mine rhodamine, can be tagged with
antibody molecules. They emit blue-green and orange-red fluorescence,
respectively under ultraviolet (UV) rays in the fluorescence microscope. This
forms the basis of the immunological test. Immunofluorescence tests have wide
applications in research and diagnostics. These tests are broadly of two types:
1.
Direct immunofluorescence test
2.
Indirect immunofluorescence test
◗ Direct immunofluorescence test
Direct immunofluorescence test is used to detect unknown antigen in
a cell or tissue by employing a known labeled anti-body that interacts directly
with unknown antigen. If antigen is present, it reacts with labeled antibody
and the antibody-coated antigen is observed under UV light of the fluorescence
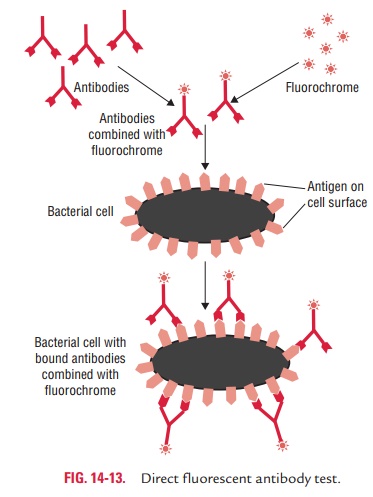
microscope (Fig. 14-13). Direct immunofluorescence test is widely used for detection of bacteria, parasites, viruses, fungi, or other antigens in CSF, blood, stool, urine, tissues, and other specimens. Few examples include:
The need for preparation of separate labeled antibody for each
pathogen is the major disadvantage of the direct immunofluo-rescence test.
◗ Indirect immunofluorescence test
The indirect immunofluorescence test is used for detection of
specific antibodies in the serum and other body fluids for sero-diagnosis of
many infectious diseases.
Indirect immunofluorescence is a two-stage process. In the first
stage, a known antigen is fixed on a slide. Then the patient’s serum to be
tested is applied to the slide, followed by careful washing. If the patient’s
serum contains antibody against the antigen, it will combine with antigen on
the slide. In the second stage, the combination of antibody with anti-gen can
be detected by addition of a fluorescent dye-labeled antibody to human IgG,
which is examined by a fluorescence microscope.
The first step in the indirect
immunofluorescence test is the incubation of a fixed antigen (e.g., in a cell
or tissue) with unlabeled antibody, which becomes associated with the antigen.
Next, after careful washing, a fluorescent antibody (e.g., fluores-cent labeled
anti-IgG) is added to the smear. This second antibody will become associated to
the first, and the antigen–antibody complex can be visualized on the
fluorescence microscope.
The indirect method has the advantage of using
a single labeled antiglobulin (antibody to IgG) as a “universal reagent” to
detect many different specific antigen–antibody reactions. The test is often
more sensitive than the direct immunofluorescence test.
The major limitation of immunofluorescence is that the tech-nique
requires (a) expensive fluorescence
microscope and reagents, (b) trained
personnel, and (c) have a factor of
subjec-tivity that may result in erroneous results.
Related Topics