Chapter: Biotechnology Applying the Genetic Revolution: Immune Technology
Humanization of Monoclonal Antibodies
HUMANIZATION
OF MONOCLONAL ANTIBODIES
Monoclonal antibodies could
theoretically be used as magic bullets to kill human cancer cells by aiming
them at specific molecules appearing only on the surface of cancer cells.
Ironically, the main problem is that the human immune system regards antibodies
from mice or other animals as foreign molecules themselves, and so attempts to
destroy them!
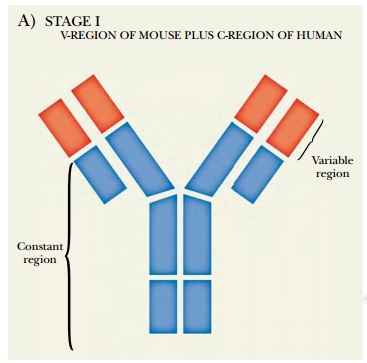
One approach that may partly
solve this problem is using genetic engineering to make humanized monoclonals (Fig. 6.9A). This takes advantage of the fact
that only the variable or V-region
of the antibody recognizes the antigen. The constant or C-region may therefore
be replaced. The first-generation hybridoma is isolated and cultured, generally
using mouse B cells. Then the DNA encoding the mouse monoclonal antibody is
isolated and cloned. The DNA for the constant region of the mouse antibody is
replaced with the corresponding human DNA sequence. The V region is left alone.
The human/mouse hybrid gene is then put back into a second mouse myeloma cell
for production of antibody in culture. Although not fully human, the hybrid is
less mouselike and provokes much less reaction from the human immune system.
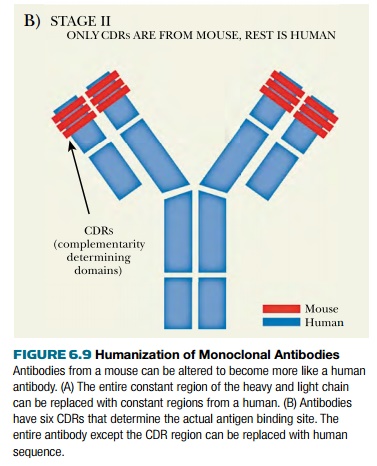
Further humanization can be
accomplished by altering those parts of the V region that are not directly
involved in binding the antigen. A closer look at the V-region of each chain
shows that most of the variation is restricted to three short segments that
form loops on the surface of the antibody, thus forming the antigen binding
site (see Fig. 6.9B). These are known as hypervariable regions or as complementarity determining regions (CDRs).
Overall, each antigen binding site consists of six CDRs, three from the light
chain and three from the heavy chain. Full humanization of an antibody involves
cutting out the coding regions for these six CDRs from the original antibody
and splicing them into the genes for human light and heavy chains.
Related Topics