Chapter: Biotechnology Applying the Genetic Revolution: Immune Technology
Antibody Structure
ANTIBODY
STRUCTURE
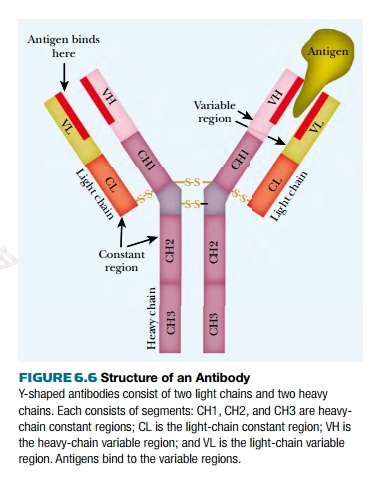
Each antibody consists of
four protein subunits, two light chains
and two heavy chains, arranged in a
Y-shape (Fig. 6.6). Disulfide bonds between cysteine amino acid residues hold
the chains together. Each of the light and heavy chains consists of a constant region and a variable region. The constant region is
the same for all chains of the same class. The variable region binds to the
target molecule, the antigen. There are millions of different variable regions,
which are generated by genetic shuffling.
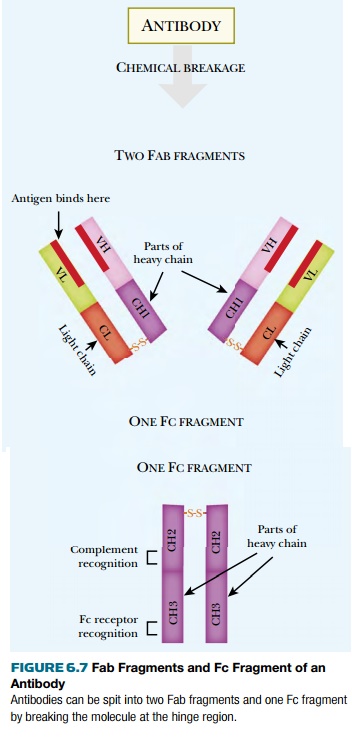
Breaking an antibody at the
“hinge” where the heavy chains bend yields three chunks, two identical Fab fragments and one Fc fragment (Fig. 6.7). Fab, meaning
“fragment, antigen binding,” consists of one light chain plus half of a heavy
chain. Fc, meaning “fragment, crystallizable,” contains the lower halves of
both heavy chains. Other components of the immune system often recognize and
bind to the Fc region of an antibody (see later discussion).
Related Topics