Chapter: Biotechnology Applying the Genetic Revolution: Immune Technology
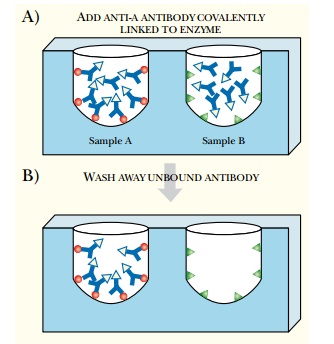
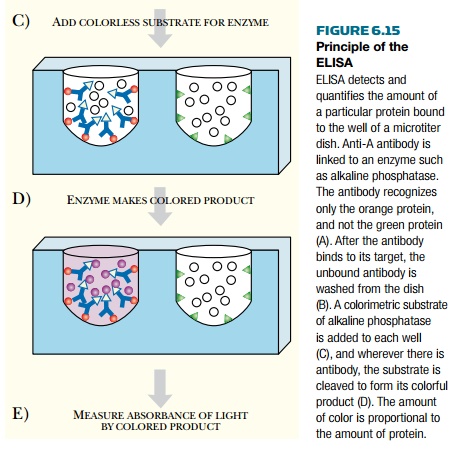
ELISA Assay
ELISA
ASSAY
The enzyme-linked
immunosorbent assay (ELISA) is widely used to detect and estimate the
concentration of a protein in a sample. The protein to be detected is regarded
as the antigen. Therefore, the first step is to make an antibody specific for
the target protein. A detection system is then attached to the rear of the
antibody. Usually this consists of an enzyme that generates a colored product
from a colorless substrate. Alkaline phosphatase, which converts X-Phos to a
blue dye, is a common choice. The samples to be assayed are immobilized on the
surface of a membrane or in the wells of a microtiter dish (Fig. 6.15). The
antibody plus detection system is added and allowed to bind. The membrane or
microtiter dish is then rinsed to remove any unbound antibody. The substrate is
added, and the intensity of color produced indicates the level of target
protein.
A variety of modifications of
the ELISA exist. Often binding and detection are done in two stages, using two
different antibodies. The first antibody is specific for the target protein.
The second antibody recognizes the first antibody and carries the detection
system. For example, antibodies could be raised in rabbits to a series of
target proteins. The second antibody, which recognizes rabbit antibodies, could
be produced in sheep. These are called secondary
antibodies and are often described
as, for example, sheep anti-rabbit. The secondary antibody has the detection system, and because it will recognize any
antibody made in a rabbit, it does not have to be reengineered for each
different target protein. This allows the use of the same final antibody
detection system in each assay even if different primary antibodies are used to
identify different proteins.
Related Topics