Chapter: Biotechnology Applying the Genetic Revolution: Immune Technology
Creating a Vaccine
CREATING
A VACCINE
Because vaccines are such a
huge part of the biotechnology industry, and such an important part of our
health care system, much research and money are invested in finding new and
improved vaccines. Many vaccines are administered to young babies; thus,
ensuring the safety and effectiveness of vaccines is critical. Many different
methods of developing a vaccine exist.
Most vaccines are simply the
disease agent, killed with high heat or denatured chemically. Heat or chemical
treatment inactivates the virus or bacterium so it cannot cause disease. Yet
enough of the original structure exists to stimulate immunity. When the live
agent infects the vaccinated person, memory B cells are activated and the
disease is suppressed. Such whole vaccines elicit the best immune response, but
many diseases cannot be isolated or cultured to make whole vaccines. Other
times, the cost of culturing the pathogen is prohibitive. Moreover, growing
live viruses is a dangerous job, with potential exposure of lab workers. With
these limitations in mind, many different strategies have been developed to
make improved vaccines.
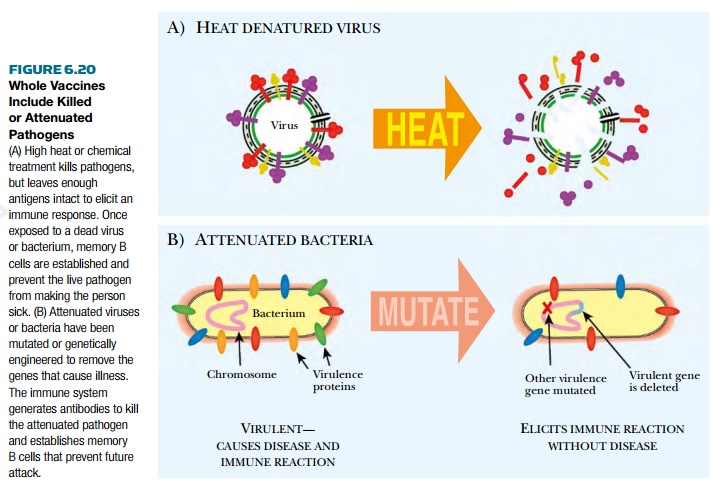
Attenuated vaccines are still-living pathogens that no longer
express the toxin or proteins that
cause the disease symptoms (Fig. 6.20). Sometimes, viruses or bacteria are
genetically engineered to remove the genes that cause disease. Other attenuated
vaccines are related but nonpathogenic strains of the infectious agent . Making attenuated virus does not pose the same risks as for live virus.
However, much research is needed to identify those genes that cause disease.
Another disadvantage is that an attenuated virus might revert to the pathogenic
version, especially if the attenuated virus was mutated in only one
disease-causing gene.
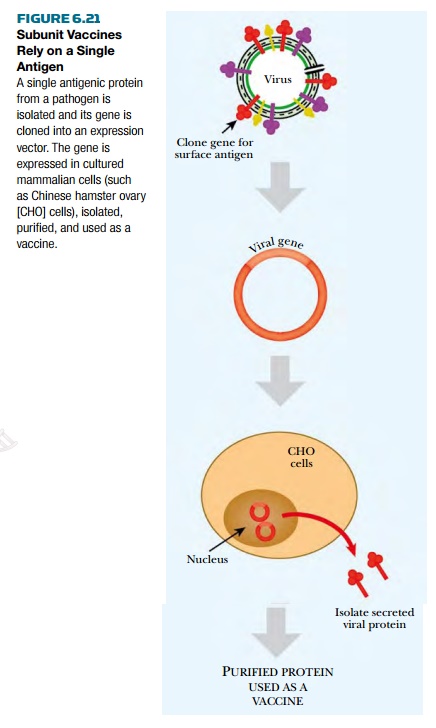
Subunit vaccines are vaccines against one component or protein of the disease agent, rather than the whole disease (Fig. 6.21). Subunit vaccines are available only because of recombinant DNA technology. The first step in creating a subunit vaccine is identifying a potential protein or part of a protein that elicits a good immune response. Interior proteins from the pathogen would not trigger the immune system when the real virus challenged it. Because of this, most subunit vaccines are made from proteins found on the outer surface of the virus or bacterium. Experiments must be done to evaluate the protein chosen for the subunit vaccine. Once a suitable protein is identified, it is expressed in cultured mammalian cells, eggs, or some other easily maintained system. The target protein is isolated from other proteins and used to immunize mice to test its effectiveness. After extensive testing in animals, the purified protein is used as a vaccine.
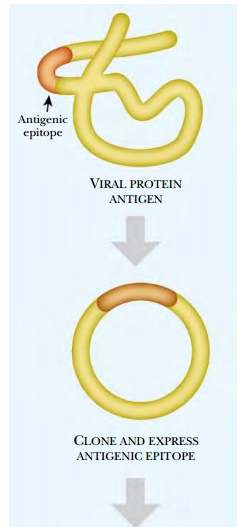
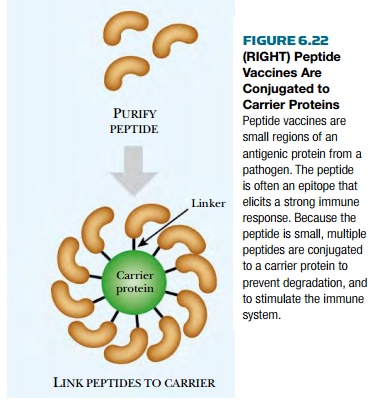
Sometime subunit vaccines fail, perhaps because the protein does not form the correct structure when expressed in mammalian cells or eggs. In these cases, peptide vaccines are created. These vaccines just use a small region of the protein. Since such peptides are small, they are conjugated to a carrier or adjuvant (Fig. 6.22). These range from live nonpathogenic viruses or bacteria, to other proteins that stimulate a better immune response than the vaccine protein itself.
Related Topics