Chapter: Basic & Clinical Pharmacology : Hypothalamic & Pituitary Hormones
Growth Hormone Antagonists
GROWTH HORMONE ANTAGONISTS
The
need for antagonists of GH stems from the tendency of GH-producing cells
(somatotrophs) in the anterior pituitary to form GH-secreting tumors. Pituitary
adenomas occur most commonly in adults. In adults, GH-secreting adenomas cause acromegaly, which is characterized by
abnormal growth of cartilage and bone tissue, and many organs including skin,
muscle, heart, liver, and the gastroin-testinal tract. Acromegaly adversely
affects the skeletal, muscular, cardiovascular, respiratory, and metabolic
systems. When a GH-secreting adenoma occurs before the long bone epiphyses
close, it leads to the rare condition, gigantism.
Small GH-secreting adenomas can be treated with GH antagonists. These include
somatostatin analogs and dopamine receptor agonists, which reduce the
production of GH, and the novel GH receptor antago-nist pegvisomant, which prevents GH from activating GH signal-ing
pathways. Larger pituitary adenomas produce greater amounts of GH and also can
impair visual and central nervous system func-tion by encroaching on nearby
brain structures. These are treated with transsphenoidal surgery or radiation.
Somatostatin Analogs
Somatostatin,
a 14-amino-acid peptide (Figure 37–2), is found in the hypothalamus, other
parts of the central nervous system, the pancreas, and other sites in the
gastrointestinal tract. It inhibits the release of GH, TSH, glucagon, insulin,
and gastrin.
Exogenous
somatostatin is rapidly cleared from the circulation, with an initial half-life
of 1–3 minutes. The kidney appears to play an important role in its metabolism
and excretion.
Somatostatin
has limited therapeutic usefulness because of its short duration of action and
multiple effects in many secretory.
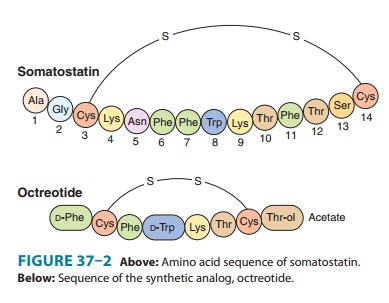
A series of longer-acting somatostatin analogs that retain biologic activity
have been developed. Octreotide, the
most widely used somatostatin analog (Figure 37–2), is 45 times more potent
than somatostatin in inhibiting GH release but only twice as potent in reducing
insulin secretion. Because of this relatively reduced effect on pancreatic beta
cells, hyperglycemia rarely occurs during treatment. The plasma elimination
half-life of octreotide is about 80 minutes, 30 times longer than that of
somatostatin.
Octreotide,
50–200 mcg given subcutaneously every 8 hours, reduces symptoms caused by a
variety of hormone-secreting tumors: acromegaly; the carcinoid syndrome;
gastrinoma; gluca-gonoma; nesidioblastosis; the watery diarrhea, hypokalemia,
and achlorhydria (WDHA) syndrome; and diabetic diarrhea. Somatostatin receptor
scintigraphy, using radiolabeled octreotide, is useful in localizing
neuroendocrine tumors having somatostatin receptors and helps predict the
response to octreotide therapy. Octreotide is also useful for the acute control
of bleeding from esophageal varices.
Octreotide
acetate injectable long-acting suspension is a slow-release microsphere
formulation. It is instituted only after a brief course of shorter-acting
octreotide has been demonstrated to be effective and tolerated. Injections into
alternate gluteal muscles are repeated at 4-week intervals in doses of 20–40 mg.
Adverse
effects of octreotide therapy include nausea, vomiting, abdominal cramps,
flatulence, and steatorrhea with bulky bowel movements. Biliary sludge and
gallstones may occur after 6 months of use in 20–30% of patients. However, the
yearly inci-dence of symptomatic gallstones is about 1%. Cardiac effects
include sinus bradycardia (25%) and conduction disturbances (10%). Pain at the
site of injection is common, especially with the long-acting octreotide
suspension. Vitamin B12 deficiency
may occur with long-term use of octreotide.
A
long-acting formulation of lanreotide,
another octapeptide somatostatin analog, was approved by the FDA in 2007 for
treat-ment of acromegaly. Lanreotide appears to have effects comparable to
those of octreotide on reducing GH levels and normalizing IGF-I concentrations.
Pegvisomant
Pegvisomant is a GH
receptor antagonist used to treat acromegaly. It is the polyethylene glycol
(PEG) derivative of a mutant GH, B2036. Like native GH, pegvisomant has two GH
receptor bind-ing sites. However, one of the pegvisomant GH receptor binding
sites has increased affinity for the GH receptor, whereas its second GH
receptor binding site has reduced affinity. This differential receptor affinity
allows the initial step (GH receptor dimerization) but blocks the
conformational changes required for signal transduc-tion. Pegvisomant is a less
potent GH receptor antagonist than is B2036, but pegylation reduces its
clearance and improves its overall clinical effectiveness. When pegvisomant was
administered subcu-taneously to 160 patients with acromegaly daily for 12
months or more, serum levels of IGF-I fell into the normal range in 97%; two
patients experienced growth of their GH-secreting pituitary tumors and two
patients developed increases in liver enzymes.
Related Topics