Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Immune Response to Infection
Functional Properties of Immunoglobulins
Functional Properties of Immunoglobulins
Immunoglobulin G Antibody
Immunoglobulin G is the most abundant immunoglobulin in health and provides the most extensive and long-lived antibody response to the various microbial and other antigens that are encountered throughout the life span of the individual. Although at least four sub-classes of IgG have been characterized. The IgG molecule is bivalent, with two identical and specific combining sites. The rest of the molecule is the constant (Fc) region, which does not vary with differences in specificity of combining sites of different antibody molecules. The constant region has specific sites for binding to phagocytic cells and for reaction with the first component of complement. These sites are made available when the variable region of the antibody molecule has reacted with specific antigen.
Immunoglobulin G antibody is characteristically formed in large amounts during the secondary response to an antigenic stimulus and usually follows production of IgM in the course of a viral or bacterial infection. Memory cells are programmed for rapid IgG response when another antigenic stimulus of the same type occurs later. Im-munoglobulin G antibodies are the most significant antibody class for neutralizing soluble antigens (eg, exotoxins) and viruses. They act by blocking the sites on the antigenic mole-cule or virus that determine attachment to cell receptors. IgG also enhances phagocytosis of particulate antigens such as bacteria, because the exposed Fc sites of antibody that is bound to the antigen have a specific affinity for receptors on the surface of phagocytic cells. As described later, the third component of complement also mediates attachment to phagocytes. Enhancement of phagocytosis by antibody, complement, or both is referred to as opsonization. Accelerated IgG responses from memory cell expansion frequently confer lifelong immunity when directed against microbial antigens that are determinants of virulence. There is active transport of the IgG molecule across the placental barrier, which allows maternal protective antibody to pass and, thus, provides passive immune protection to the fetus and newborn pending development of a mature immune system. It is the only immunoglobulin class known to be placentally transferred. The half-life of passively transferred IgG within the same species is approximately 1 month, and thus the infant is protected during a particularly vulnerable period of life.
Immunoglobulin M Antibody
Monomers of IgM constitute the specific epitope recognition sites on B cells that ulti-mately give rise to plasma cells producing one or another of the different immunoglobulin classes of antibody. Because of its multiple specific combining sites, IgM is particularly effective in agglutinating particles carrying epitopes against which it is directed. It also contains multiple sites for binding the first component of complement. These sites be-come available once the IgM molecule has reacted with antigen. IgM is particularly ac-tive in bringing about complement-mediated cytolytic damage to foreign antigen-bearing cells. It is not, itself, an opsonizing antibody because its Fc portion is not recognized by phagocytes. Opsonization occurs through its activation of the complement pathway.
Immunoglobulin M is usually the earliest antibody to appear after an antigenic stimu-lus, but it tends to decline rapidly and is often succeeded by IgG production from the same clone of cells. It is primarily intravascular and does not cross the placental barrier to the fetus (in contrast to IgG). Thus, the presence of specific IgM against a potentially in-fecting agent in the blood of a neonate is a priori evidence of active infection rather than of passively acquired antibody from the mother. Antibody response to certain antigens, including the lipopolysaccharide O antigen of Gram-negative bacteria, is characteristi-cally IgM. Some universally occurring antibodies (natural antibodies), such as those di-rected against blood group antigens, are also of the IgM class.
Immunoglobulin A Antibody
Immunoglobulin A has a special role as a major determinant of so-called local immunity in protecting epithelial surfaces from colonization and infection. Certain B cells in lym-phoid tissues adjacent to or draining surface epithelia of the intestines, respiratory tract, and genitourinary tract are encoded for specific IgA production. After antigenic stimulus, the clone expands locally and some of the IgA-producing cells also migrate to other vis-cera and secretory glands. At the epithelia, two IgA molecules combine with another pro-tein, termed thesecretory piece, which is present on the surface of local epithelial cells. The complex, then termed secretory IgA (sIgA), passes through the cells into the mucous layer on the epithelial surface or into glandular secretions where it exerts its protective ef-fect. The secretory piece not only mediates secretion but also protects the molecule against proteolysis by enzymes such as those present in the intestinal tract.
The major role of sIgA is to prevent attachment of antigen-carrying particles to receptors on mucous membrane epithelia. Thus, in the case of bacteria and viruses, it reacts with sur-face antigens that mediate adhesion and colonization and prevents the establishment of local infection or invasion of the subepithelial tissues. It can agglutinate particles but has no Fc do-main for activating the classic complement pathway; however, it can activate the alternative pathway . Reaction of IgA with antigen within the mucous membrane initiates an inflammatory reaction that helps mobilize other immunoglobulin and cellular defenses to the site of invasion. IgA response to an antigen is shorter lived than the IgG response.
Immunoglobulin E Antibody
Immunoglobulin E is a monomer consisting of two light chains (either k or l ) and two heavy chains. It is normally present in very small amounts in serum, and most IgE is bound firmly by its Fc portion to tissue mast cells and basophils, which are major producers of histamine. When IgE bound to mast cells reacts with specific antigen, the mast cells degranulate and release histamine and other factors that mediate an inflammatory reaction with dilation of the capillaries, exudation of plasma components, and attraction of neutrophils and eosinophils to the site. Thus, IgE contributes to a rapid second line of defense if surface-protective mechanisms are breached. IgE also plays a significant indirect role in the immune response to a number of helminthic (worm) infections because of attraction of eosinophils to the site at which it reacts with antigen. The eosinophils bind to the Fc portions of IgG molecules that have reacted with surface antigens of the parasite and help bring about its destruction. Certain types of allergies, to be discussed later, are due to excessive production of IgE with specificity for a foreign protein. The pharmacologic effects of histamine and the other vasoactive mediators released from mast cells largely account for the symptoms of the disorder.
Immunoglobulin D Antibody
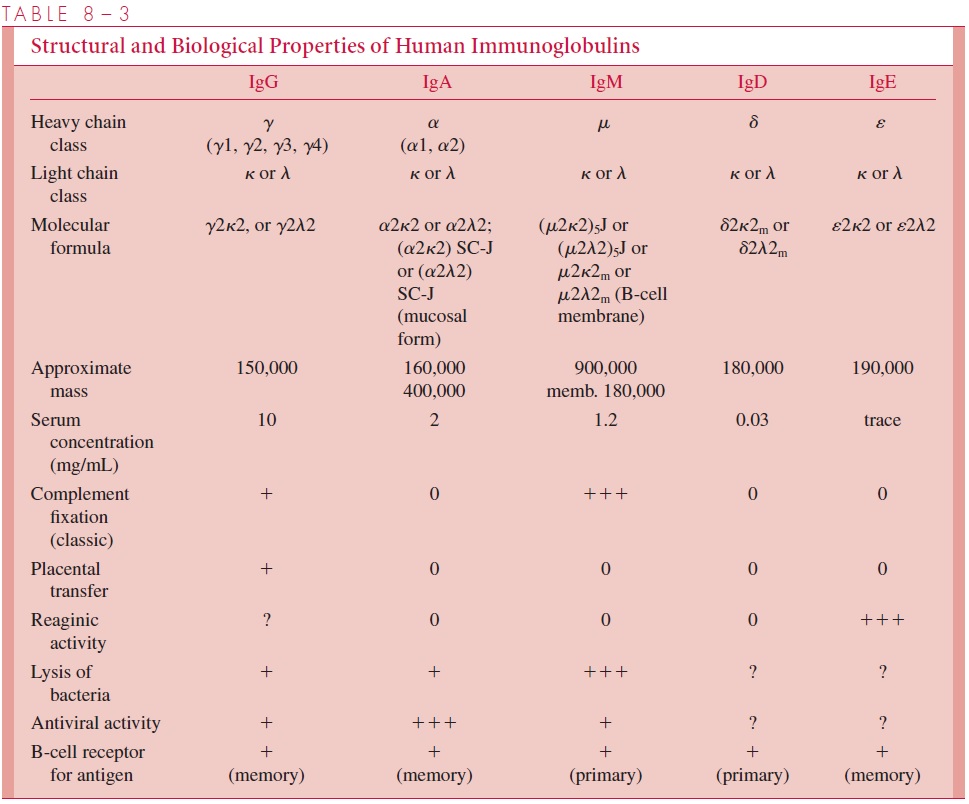
Immunoglobulin D antibody consists of two light chains and two heavy chains. It is highly susceptible to proteolytic enzymes in the tissues and is found only in very low concentrations in serum. Its role is not fully understood, although, as indicated earlier, it is present on the surface of unstimulated B cells and may serve as a receptor for antigen. The chain composition, size, and some major biological properties of the separate classes of immunoglobulins are summarized in Table 8 – 3.
Related Topics