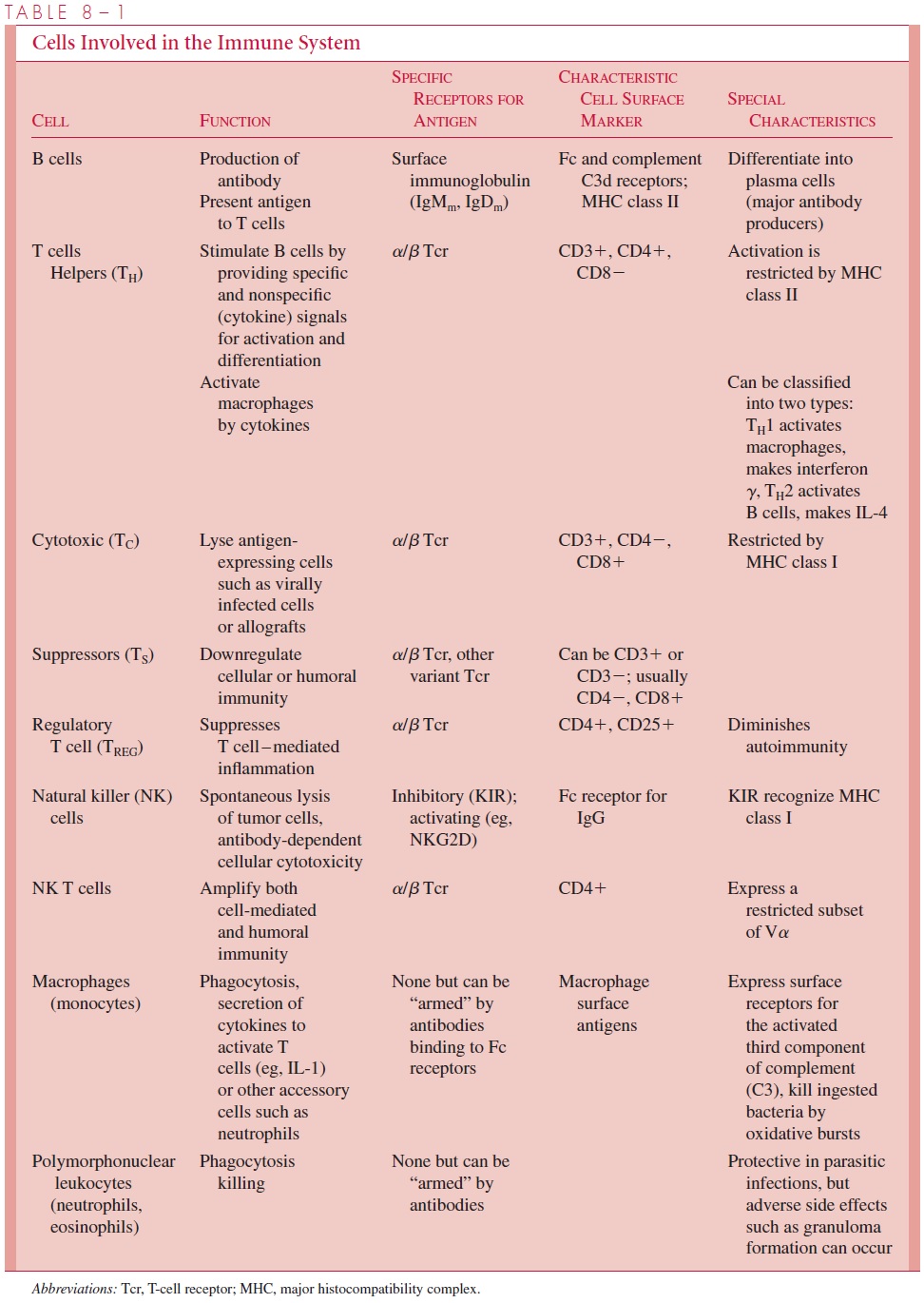
Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Immune Response to Infection
Adverse Effects of Immunologic Reactions and Hypersensitivity
ADVERSE EFFECTS OF IMMUNOLOGIC REACTIONS AND HYPERSENSITIVITY
Immunologic reactions in the body may result in excessive responses, sometimes far be- yond those needed to remove or neutralize microbial pathogens or molecules contributing to disease. Such responses are classified as hypersensitivity reactions if they cause marked physiologic changes or tissue damage or exacerbate disease processes. Four dis- tinct classes of hypersensitivity are recognized, but these do not occur in isolation, and in- jury often results from a combination of the reactions. In each case they represent an extension of a normal defense mechanism. The immune response in practice is very potent and leads to deleterious consequences for the host if it is in operation too long. The antigen-specific portion of the humoral or T-cell reactions constitutes only a small frac- tion of the overall response and amplification by complement components that can stimu- late phagocytic cells and the release of cytokines; these can cause a general recruitment of lymphocytes, monocytes, and polymorphonuclear cells, leading to a cascade of inflam- mation and prolonged disease. The aspects of hypersensitivity are overexpressions of the beneficial immune responses that act inappropriately. The normal immune basis of the four types of hypersensitivity are described below and included in Tables 8 – 1 and 8 – 4.
Types I, II, and III hypersensitivity are mediated by antibody; type IV (delayed-type) hypersensitivity is carried out by antigen-specific T cells assisted by macrophages.
Anaphylactic (Type I) Hypersensitivity
Type I hypersensitivity, also called anaphylaxis, is represented by the allergic reactions that occur immediately following contact with the sensitizing antigen (allergen). IgE anti-bodies are bound to Fc receptors on the surface of mast cells (Fig 8 – 10). If a multivalent antigen binds to the cell-bound IgE molecules, it cross-links them, with the result that the mast cell degranulates, releasing a variety of pharmacologically active mediators. Among the prominent mediators released following binding of the allergen is histamine, which increases capillary permeability and causes bronchoconstriction. Preformed mediators such as the anticoagulant heparin, complement 3 convertase, and a group of compounds involved in chemotaxis of eosinophils, neutrophils, and platelet activation are also re-leased by degranulation. Slow reactive substances involved in bronchoconstriction and

chemotaxis are also produced, as are prostaglandins and thromboxanes, which are impli-cated in bronchospasm, muscle contraction, and platelet aggregation. Thus, the specific binding of an allergen to the combining site of its antibody can result in a potent release of compounds, leading to painful and life-threatening consequences for the allergic indi-vidual. Despite the suffering that hypersensitivity to common allergens such as pollen, bee stings, house dust, and cat dander brings to a large percentage of people, there are possible beneficial consequences of binding of allergen by IgE. These include situations in which ADCC by monocytes and eosinophils may provide protection against parasites such as schistosomes (a trematode worm) and trypanosomes (a protozoan).
When hypersensitivity is very marked, or antigen is introduced systemically, mast cells throughout the body degranulate, and systemic anaphylaxis results with constriction of the bronchi, edema of the larynx and other tissues, vascular collapse, and sometimes death. A generalized anaphylactic reaction rarely if ever occurs as a manifestation of an infection but may occur following parenteral inoculation of an antigen to which the individual has been sensitized (eg, bee sting venom). It may also occur in individuals who have been sensitized to a low-molecular-weight hapten that binds to a tissue protein and becomes antigenic because of the size of the complex. An IgE response to hapten epitopes can then lead to anaphylactic-type hypersensitivity if the epitope is again encountered. A penicillin degradation product has this property, and occasionally, individuals develop severe ana-phylactic reactions to penicillins, although this complication is very rare.
Rapid therapeutic intervention is critical in systemic anaphylaxis. It includes parenteral administration of epinephrine, which reverses the major manifestations of the syndrome by producing bronchodilation, vasoconstriction, and increased blood pressure. Tracheostomy or intubation may be needed to overcome respiratory obstruction due to laryngeal edema.
Antibody-Mediated (Type II) Hypersensitivity
Type II hypersensitivity is an inappropriate elaboration of antibody-dependent cytotoxic-ity that occurs when antibody binds to antigens on host cells, leading to phagocytosis, killer cell activity, or complement-mediated lysis. Antibody directed against cell surface or tissue antigens results in the fixation of complement such that a variety of effector cells become involved. The cells to which the antibody is specifically bound, as well as the surrounding tissues, are damaged because of the inflammatory amplification. Such mech-anisms appear to be responsible for the tissue damage of rheumatic fever following a streptococcal infection or some clinical manifestations of viral diseases, such as group B coxsackievirus infection . These phenomena may involve not only antibodies but also cytotoxic T cells. It should be recalled that humoral antibodies are required to arm macrophages, and PMNs are needed to bind cellular antigens in ADCC and to serve as opsonins that facilitate ingestion with eventual intracellular destruction of target cells by macrophages.
Immune Complex (Type III) Hypersensitivity
When IgG is mixed in appropriate proportions with multivalent antigen molecules (ie, bearing multiple epitopes), aggregates containing a lattice of many antigen and antibody molecules forms. With appropriate concentrations of the two reactants, a macroscopic precipitate can develop . A similar situation applies to IgM, which is multivalent. When the epitope is present on the surface of a larger particle, such as a bac-terium or red blood cell, the particles can be cross-linked by antibody, and microscopic or macroscopic agglutination results. These phenomena can occur in vivo when sufficient amounts of specific antibody and of free antigen from an infecting microorganism react locally or in the bloodstream to form an antigen – antibody lattice; the size of the immune complex depends on the relative properties of the two reactants. Large immune com-plexes are phagocytosed and usually broken down within the phagocyte. However, smaller complexes are deposited in small blood vessels and capillaries through which they do not pass, activate the complement system, and thus produce an acute inflamma-tory response mediated largely by C3a and C5a. This results in the manifestations of vasculitis. Phagocytes attracted chemotactically to the site release hydrolytic enzymes, and the sum of these effects is acute tissue damage, which can become chronic depending on the survival of the antigen or on whether it is continually replaced. Acute glomeru-lonephritis following certain streptococcal infections is an example of an immune com-plex disease in which glomeruli of the kidney are damaged by the complexes, resulting in various manifestations of renal impairment. Inflammatory skin lesions can result from deposition of immune complexes in the cutaneous blood vessels in patients with infective endocarditis. Deposition in joints, the pericardium, or the pleura produces arthritis, peri-carditis, and pleuritis or pleurisy, respectively.
A systemic form of immune complex disease, termed serum sickness, can follow the injection of foreign antigen. An example is the therapeutic use of diphtheria antitoxin that has been produced in horses. About 10 days after inoculation, sufficient antibody against horse proteins has been produced to form immune complexes made up of human anti-body reacting against horse serum protein (including horse immunoglobulins). These complexes are deposited in various organs, resulting in a syndrome of arthritis, nephritis, rash, urticaria, and fever. The disease usually resolves as the foreign antigen concentra-tion decreases through immune clearance and catabolism of the antigen(s).
Delayed-Type (Type IV) Hypersensitivity
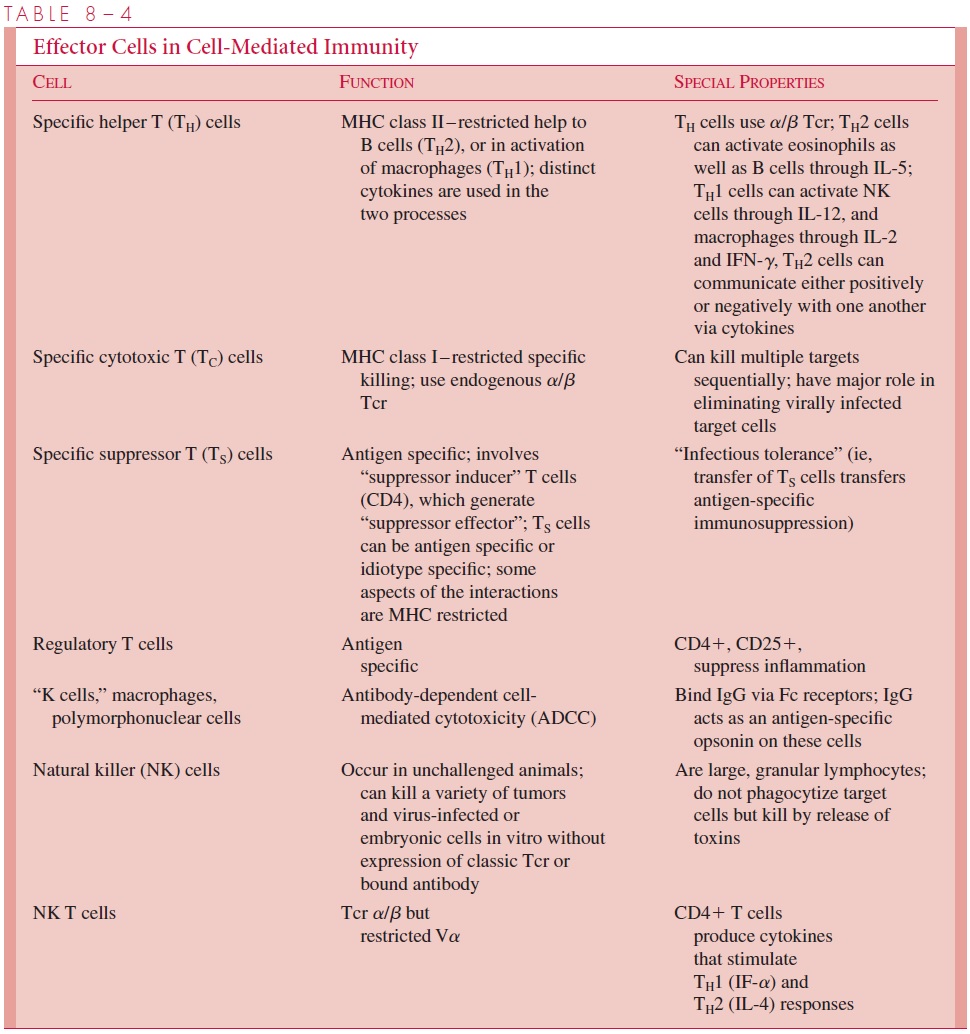
The fourth type of hypersensitivity is termed, delayed-type hypersensitivity. Unlike types I, II, and III, this process cannot be transferred from one animal to another by serum alone. However, it can be transferred by antigen-specific T lymphocytes. All are initiated by the function of antigen-specific T cells, which then recruit effector cells into the area of recognition of the antigen. Unlike the forms of “immediate” hypersensitivity that can be transferred by antibody, delayed-type hypersensitivity requires days to weeks to express full reactivity. In all of these, the initial reaction is the induction and function of antigen-specific T cells that bear α/β T-cell receptors and have been generated in re-sponse to antigenic challenge. The time course depends on the involvement of other cells and the properties of the infectious agent involved.
Four major types of delayed-type hypersensitivity are all part of the same process dif-fering in site, mechanism of challenge, and timing. The shortest is the Jones – Mote phe-nomenon, in which the site of antigen injection is infiltrated by basophils, and the skin swelling is maximal 24 hours after antigen injection. This type of hypersensitivity can be raised to soluble antigens, and the reactivity disappears following the appearance of anti-body. Contact and tuberculin-type hypersensitivity show maximal reactivity at 48 to 72 hours. Contact sensitivity is observed in response to sensitization with common antigens such as chemicals found in rubber or the small organic compounds produced by poison ivy and poison oak. It is predominantly an epidermal reaction, in contrast to the tuber-culin-type hypersensitivity, which is a dermal reaction. The cell that presents antigen for contact sensitization is the Langerhans cell, a dendritic antigen-presenting cell carrying MHC class II antigens. Tuberculin-type hypersensitivity is manifested by individuals who have been sensitized with lipoprotein antigens derived from the tubercle bacillus. Twenty-four hours after intradermal injection of tuberculin, an antigen derived from Mycobacterium tuberculosis, there is intense infiltration by lymphocytes, which reaches amaximum in 2 to 3 days.
Probably the most clinically important form of type IV sensitivity is the granuloma, an organized inflammatory lesion that requires at least 14 days to develop. These result from the long-term continuation of the stimulation of effector cells by cytokines produced in initial antigen-specific T-cell response. Granulomatous lesions are a major part of the disease process in chronic diseases caused by bacteria (tuberculosis), fungi (histoplasmo-sis), and parasites (schistosomiasis).
Related Topics