Chapter: Biology of Disease: Toxicology
Ethylene Glycol - Toxicology Poisons
ETHYLENE GLYCOL
The alcohol ethylene glycol is a commonly used component of antifreeze,
paints, polishes and cosmetics. It is a poison with a minimum lethal dose of
approximately 100 cm3. The toxicity of ethylene glycol is due to its
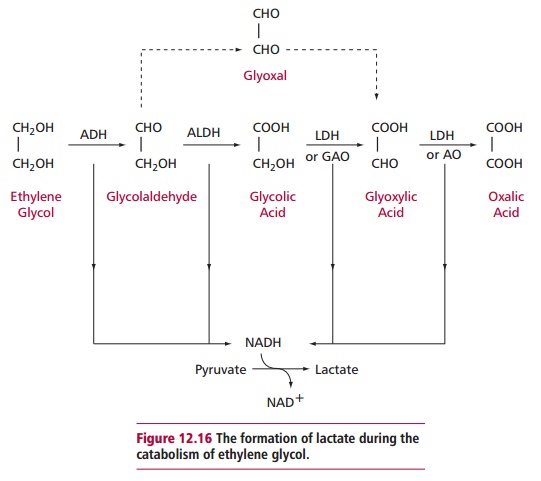
oxidation to oxalate, which humans cannot rapidly excrete. The oxidative steps
produce excessive NADH resulting in lactate production (Figure 12.16). The oxalate and lactate can result in metabolic
acidosis, while the organic acids produced by the breakdown of ethylene glycol
inhibit a number of metabolic processes
including oxidative phosphorylation. The production of oxalate
also results in its deposition as insoluble calcium oxalate in renal tubules
and the brain. These biochemical events reduce the concentrations of hydrogen
carbonate and Ca2+, but increase that of K+ in plasma.
Crystals, blood and protein may all leak into the urine, giving rise to three
distinct clinical phases. Within 30 min to 12 h, intoxication, nausea,
vomiting, coma, convulsions, nystagmus, papilloedema, depressed reflexes,
myclonic jerks, tetanic contractions and permanent optic atrophy may occur. In
12 to 23 h tachypnea, tachycardia, hypertension, pulmonary edema and congestive
cardiac failure may all present. In the following 24 to 72 h kidney damage with
pain and acute renal tubular necrosis may feature. Death may occur within 24 h
due to damage to the CNS, or between eight to 12 days due to renal failure.
Treatment of ethylene glycol poisoning is to apply gastric
lavage to reduce its absorption, combined with supportive therapies for the
shock and respiratory distress. Administration of ethanol is standard since it
competes effectively for the active site on alcohol dehydrogenase, inhibiting
the metabolism of the absorbed ethylene glycol. Sodium hydrogen carbonate,
administered intravenously, and calcium gluconate are used to correct the
acidosis and hypocalcemia respectively. Dialysis is also used to remove ethylene
glycol.
Related Topics