Chapter: Clinical Anesthesiology: Perioperative & Critical Care Medicine: Management of Patients with Fluid & Electrolyte Disturbances
Disorders of Water Balance: Hyperosmolality & Hypernatremia
HYPEROSMOLALITY & HYPERNATREMIA
Hyperosmolality occurs whenever total body
solute content increases relative to TBW and is usually, but not always,
associated with hypernatremia ([Na+]145 mEq/L). Hyperosmolality without
hyperna-tremia may be seen during marked hyperglycemia or following the
accumulation of abnormal osmoti-cally active substances in plasma (see above).
In the latter two instances, plasma sodium concentration may actually decrease
as water is drawn from the intracellular to the extracellular compartment. For
every 100 mg/dL increase in plasma glucose con-centration, plasma sodium
decreases approximately 1.6 mEq/L.
Hypernatremia is nearly always the result of either a relative loss of
water in excess of sodium (hypotonic fluid loss) or the retention of large
quan-tities of sodium. Even when renal concentrating ability is impaired,
thirst is normally highly effec-tive in preventing hypernatremia. Hypernatremia
is therefore most commonly seen in debilitated patients who are unable to
drink, the very aged, the very young, and patients with altered con-sciousness.
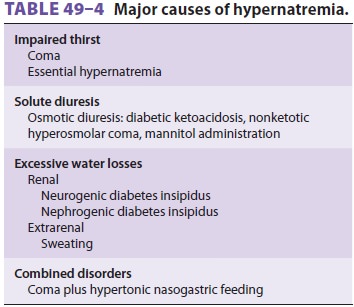
Patients with hypernatremia may have a low, normal, or high total body sodium
content (Table 49–4).
Hypernatremia & Low Total Body Sodium Content
These patients have lost both sodium and water, but the water loss is in relative excess to that of the sodium loss. Hypotonic losses can be renal (osmotic diuresis) or extrarenal (diarrhea or sweat). In either case, patients usually manifest signs of hypovolemia . Urinary sodium concentration is generally greater than 20 mEq/L with renal losses and less than 10 mEq/L with extrarenal losses.
Hypernatremia & Normal Total Body Sodium Content
This group of patients generally manifests
signs of water loss without overt hypovolemia unless the water loss is massive.
Total body sodium content is generally normal. Nearly pure water losses can
occur via the skin, respiratory tract, or kidneys. Occasionally transient
hypernatremia is observed with movement of water into cells following
exer-cise, seizures, or rhabdomyolysis. The most com-mon cause of hypernatremia
in conscious patients with normal total body sodium content is diabetesinsipidus. Diabetes insipidus is characterized bymarked impairment in renal
concentrating ability that is due either to decreased ADH secretion (cen-tral
diabetes insipidus) or failure of the renal tubules to respond normally to
circulating ADH (nephro-genic diabetes insipidus). Rarely, “essential
hyperna-tremia” may be encountered in patients with central nervous system
disorders. These patients appear to have “reset” osmoreceptors that function at
a higher baseline osmolality.
A. Central Diabetes Insipidus
Lesions in or around the hypothalamus and the
pituitary stalk frequently produce diabetes insipi-dus. Diabetes insipidus
often develops with brain death. Transient diabetes insipidus is also com-monly
seen following neurosurgical procedures and head trauma. The diagnosis is
suggested by a his-tory of polydipsia, polyuria (often >6 L/d), and the absence of hyperglycemia. In
the perioperative set-ting, the diagnosis of diabetes insipidus is suggested by
marked polyuria without glycosuria and a uri-nary osmolality lower than plasma
osmolality. The absence of thirst in unconscious individuals leads to marked
water losses and can rapidly produce hypo-volemia. The diagnosis of central
diabetes insipidus is confirmed by an increase in urinary osmolality following
the administration of exogenous ADH. Aqueous vasopressin (5–10 units
subcutaneously or intramuscularly every 4–6 h) is the treatment of choice for
acute central diabetes insipidus. Vaso-pressin in oil (0.3 mL intramuscularly
every day) is longer lasting but is more likely to cause water intoxication.
Desmopressin (DDAVP), a synthetic analogue of ADH with a 12- to 24-h duration
of action, is available as an intranasal preparation (10–40 mcg/d either as a
single daily dose or divided into two doses) that can be used in both
ambulatory and perioperative settings.
B. Nephrogenic Diabetes Insipidus
Nephrogenic diabetes insipidus can be congenital but is more commonly
secondary to other dis-orders, including chronic kidney disease, hypo-kalemia
and hypercalcemia, sickle cell disease, and hyperproteinemias. Nephrogenic
diabetes insipidus can also be secondary to the side effects of some drugs
(amphotericin B, lithium, dem-eclocycline, ifosfamide, mannitol). ADH
secre-tion in nephrogenic diabetes insipidus is normal, but the kidneys fail to
respond to ADH; urinary concentrating ability is therefore impaired. The
mechanism may be either a decreased response to circulating ADH or interference
with the renal countercurrent mechanism. The diagnosis is confirmed by failure
of the kidneys to produce hypertonic urine following the administration of
exogenous ADH. Treatment is generally directed at the underlying illness and
ensuring an ade-quate fluid intake. Volume depletion by a thia-zide diuretic
can paradoxically decrease urinary output by reducing water delivery to
collecting tubules. Sodium and protein restriction can simi-larly reduce
urinary output.
Hypernatremia & Increased Total Body Sodium Content
This condition most commonly results from the administration of large
quantities of hypertonic saline solutions (3% NaCl or 7.5% NaHCO 3).
Patients with primary hyperaldosteronism and Cushing’s syndrome may also have
elevations in serum sodium concentration along with signs of increased sodium
retention.
Clinical Manifestations of Hypernatremia
Neurological manifestations predominate in
patients with hypernatremia and are generally thought to result from cellular
dehydration. Restlessness, leth-argy, and hyperreflexia can progress to
seizures, coma, and ultimately death. Symptoms correlate more closely with the
rate of movement of water out of brain cells than with the absolute level of
hyper-natremia. Rapid decreases in brain volume can rup-ture cerebral veins and
result in focal intracerebral or subarachnoid hemorrhage. Seizures and serious
neurological damage are common, particularly in children with acute
hypernatremia when plasma [Na+] exceeds 158 mEq/L. Chronic hypernatremia is
usually better tolerated than the acute form. After 24–48 h, intracellular
osmolality begins to rise as a result of increases in intracellular inositol
and amino acid (glutamine and taurine) concentrations. As intracellular solute
concentration increases, neuro-nal water content slowly returns to normal.
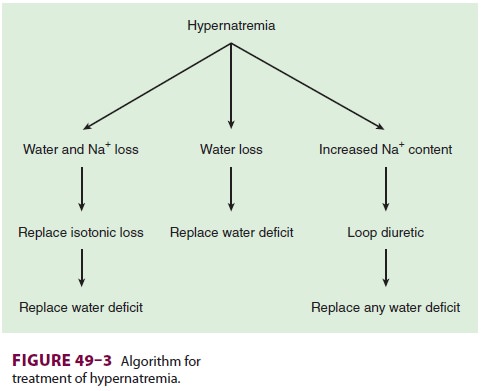
Treatment of Hypernatremia
The treatment of hypernatremia is aimed at restor-ing plasma osmolality
to normal as well as cor-recting the underlying cause. Water deficits should
generally be corrected over 48 h with a hypotonic solution such as 5% dextrose
in water . Abnormalities in extracellular volume must also be corrected (Figure
49–3). Hypernatremic patients with decreased total body sodium should be
given isotonic fluids to restore plasma volume to normal prior to treatment with a hypotonic solution. Hyper-natremic
patients with increased total body sodium should be treated with a loop
diuretic along with intravenous 5% dextrose in water. The treatment of diabetes
insipidus is discussed above.
Rapid correction of hypernatremia can result in seizures, brain edema,
permanent neurological damage, and even death. Serial Na+ osmolalities should be
obtained during treatment. In general, decreases in plasma sodium concentration
should not proceed at a rate faster than 0.5 mEq/L/h.
Example
A 70-kg man is found to have a plasma [Na+] of 160 mEq/L. What is his water deficit?
If one
assumes that hypernatremia in this cases represents water loss only, then total
body osmoles are unchanged. Thus, assuming a normal [Na+] of 140 mEq/L and TBW content that is 60% of body weight:
Normal TBW
× 140 = present TBW × [Na+]plasma or (70 × 0.6) × 140 = present TBW × 160
Solving
the equation:
Present
TBW = 36.7 L
Water
deficit = normal TBW − present TBW or (70 × 0.6) − 36.7 = 5.3 L
To replace
this deficit over 48 h, it is necessary to give 5% dextrose in water
intravenously, 5300 mL over 48 h, or 110 mL/h.
Note that
this method ignores any coexisting isotonic fluid deficits, which if present
should be replaced with an isotonic solution.
Anesthetic Considerations
Hypernatremia has been demonstrated to increase
the minimum alveolar concentration for inhalation anesthetics in animal
studies, but its clinical signifi-cance is more closely related to the
associated fluid deficits. Hypovolemia accentuates any vasodilation or cardiac
depression from anesthetic agents and predisposes to hypotension and
hypoperfusion of tissues. Decreases in the volume of distribution for drugs
necessitate dose reductions for most intrave-nous agents, whereas decreases in
cardiac output enhance the uptake of inhalation anesthetics.
Elective surgery should be postponed in
patients with significant hypernatremia (>150 mEq/L) until the cause is established and fluid deficits are
cor-rected. Both water and isotonic fluid deficits should be corrected prior to
elective surgery.
Related Topics