Chapter: Basic & Clinical Pharmacology : Agents Used in Cardiac Arrhythmias
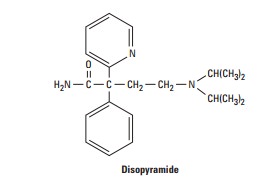
Disopyramide (SubGroup 1A)
DISOPYRAMIDE (SUBGROUP 1A)
Cardiac Effects
The
effects of disopyramide are very similar to those of procain-amide and quinidine.
Its cardiac antimuscarinic effects are even more marked than those of
quinidine. Therefore, a drug that slows AV conduction should be administered
with disopyramide when treating atrial flutter or fibrillation.
Toxicity
Toxic
concentrations of disopyramide can precipitate all of the electrophysiologic
disturbances described under quinidine. As a result of its negative inotropic
effect, disopyramide may precipi-tate heart failure de novo or in patients with
preexisting depression of left ventricular function. Because of this effect,
disopyramide is not used as a first-line antiarrhythmic agent in the USA. It
should not be used in patients with heart failure.
Disopyramide’s
atropine-like activity accounts for most of its symptomatic adverse effects:
urinary retention (most often, but not exclusively, in male patients with
prostatic hyperplasia), dry mouth, blurred vision, constipation, and worsening
of preexisting glaucoma. These effects may require discontinuation of the drug.
Pharmacokinetics & Dosage
In
the USA, disopyramide is only available for oral use. The typi-cal oral dosage
of disopyramide is 150 mg three times a day, but up to 1 g/d has been used. In
patients with renal impairment, dos-age must be reduced. Because of the danger
of precipitating heart failure, loading doses are not recommended.
Therapeutic Use
Although
disopyramide has been shown to be effective in a variety of supraventricular
arrhythmias, in the USA it is approved only for the treatment of ventricular
arrhythmias.
Related Topics