Chapter: Medical Physiology: Urine Formation by the Kidneys: I. Glomerular Filtration, Renal Blood Flow, and Their Control
Determinants of the GFR
Determinants of the GFR
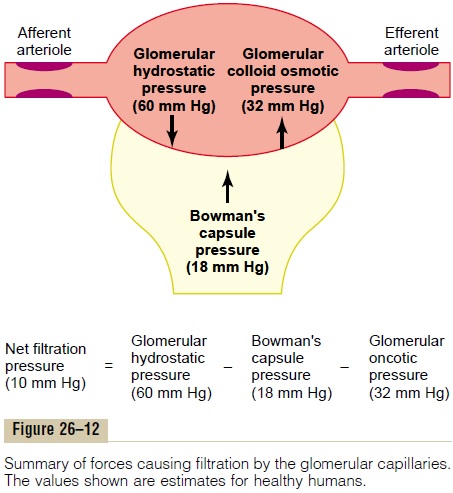
The GFR is determined by (1) the sum of the hydro-static and colloid osmotic forces across the glomerular membrane, which gives the net filtration pressure, and (2) the glomerular capillary filtration coefficient, Kf. Expressed mathematically, the GFR equals the product of Kf and the net filtration pressure:
GFR = Kf x Net filtration pressure
The net filtration pressure represents the sum of the hydrostatic and colloid osmotic forces that either favor or oppose filtration across the glomerular capillaries (Figure 26–12). These forces include (1) hydrostatic pressure inside the glomerular capillaries (glomerular hydrostatic pressure, PG), which promotes filtration; (2) the hydrostatic pressure in Bowman’s capsule (PB) outside the capillaries, which opposes filtration; (3) the colloid osmotic pressure of the glomerular capillary plasma proteins (πG), which opposes filtration; and (4) the colloid osmotic pressure of the proteins in Bowman’s capsule (πB), which promotes filtration. (Under normal conditions, the concentration of protein in the glomerular filtrate is so low that the colloid osmotic pressure of the Bowman’s capsule fluid is considered to be zero.)
The GFR can therefore be expressed as
GFR = Kf x (PG – PB – pG + pB)
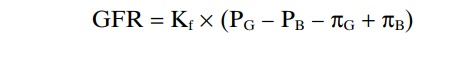
Although the normal values for the determinants of GFR have not been measured directly in humans, they have been estimated in animals such as dogs and rats. Based on the results in animals, the approximate normal forces favoring and opposing glomerular fil-tration in humans are believed to be as follows (see Figure 26–12):

Some of these values can change markedly under different physiologic conditions, whereas others are altered mainly in disease states, as discussed later.
Increased Glomerular Capillary Filtration Coefficient Increases GFR
The Kf is a measure of the product of the hydraulic conductivity and surface area of the glomerular capil-laries. The Kf cannot be measured directly, but it is estimated experimentally by dividing the rate of glomerular filtration by net filtration pressure:
Kf = GFR/Net filtration pressure
Because total GFR for both kidneys is about 125 ml/ min and the net filtration pressure is 10 mm Hg, the normal Kf is calculated to be about 12.5 ml/min/mm Hg of filtration pressure. When Kf is expressed per 100 grams of kidney weight, it averages about 4.2 ml/min/mm Hg, a value about 400 times as high as the Kf of most other capillary systems of the body; the average Kf of many other tissues in the body is only about 0.01 ml/min/mm Hg per 100 grams. This high Kf for the glomerular capillaries contributes tremen-dously to their rapid rate of fluid filtration.
Although increased Kf raises GFR and decreased Kf reduces GFR, changes in Kf probably do not provide a primary mechanism for the normal day-to-day regu-lation of GFR. Some diseases, however, lower Kf by reducing the number of functional glomerular capil-laries (thereby reducing the surface area for filtration) or by increasing the thickness of the glomerular capillary membrane and reducing its hydraulic con-ductivity. For example, chronic, uncontrolled hyper-tension and diabetes mellitus gradually reduce Kf by increasing the thickness of the glomerular capillary basement membrane and, eventually, by damaging the capillaries so severely that there is loss of capillary function.
Increased Bowman’s Capsule Hydrostatic Pressure Decreases GFR
Direct measurements, using micropipettes, of hydro-static pressure in Bowman’s capsule and at different points in the proximal tubule suggest that a reasonable estimate for Bowman’s capsule pressure in humans is about 18 mm Hg under normal conditions. Increasing the hydrostatic pressure in Bowman’s capsule reduces GFR, whereas decreasing this pressure raises GFR. However, changes in Bowman’s capsule pressure nor-mally do not serve as a primary means for regulating GFR.
In certain pathological states associated with obstruction of the urinary tract, Bowman’s capsule pressure can increase markedly, causing serious reduc-tion of GFR. For example, precipitation of calcium or of uric acid may lead to “stones” that lodge in the urinary tract, often in the ureter, thereby obstructing outflow of the urinary tract and raising Bowman’s capsule pressure. This reduces GFR and eventually can damage or even destroy the kidney unless the obstruction is relieved.
Increased Glomerular Capillary Colloid Osmotic Pressure Decreases GFR
As blood passes from the afferent arteriole through the glomerular capillaries to the efferent arterioles, the plasma protein concentration increases about 20 per cent (Figure 26–13). The reason for this is that about one fifth of the fluid in the capillaries filters into Bowman’s capsule, thereby concentrating the glomerular plasma proteins that are not filtered. Assuming that the normal colloid osmotic pressure of plasma entering the glomerular capillaries is 28 mm Hg, this value usually rises to about 36 mm Hg
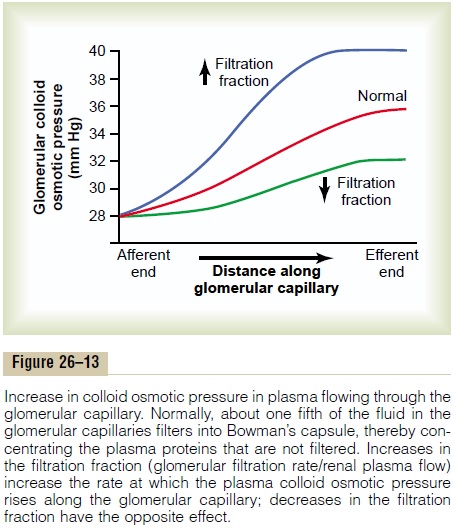
by the time the blood reaches the efferent end of the capillaries. Therefore, the average colloid osmotic pressure of the glomerular capillary plasma proteins is midway between 28 and 36 mm Hg, or about 32 mm Hg.
Thus, two factors that influence the glomerular cap-illary colloid osmotic pressure are (1) the arterial plasma colloid osmotic pressure and (2) the fraction of plasma filtered by the glomerular capillaries (filtra-tion fraction). Increasing the arterial plasma colloid osmotic pressure raises the glomerular capillary colloid osmotic pressure, which in turn decreases GFR.
Increasing the filtration fraction also concentrates the plasma proteins and raises the glomerular colloid osmotic pressure (see Figure 26–13). Because the fil-tration fraction is defined as GFR/renal plasma flow, the filtration fraction can be increased either by raising GFR or by reducing renal plasma flow. For example, a reduction in renal plasma flow with no initial change in GFR would tend to increase the filtration fraction, which would raise the glomerular capillary colloid osmotic pressure and tend to reduce GFR. For this reason, changes in renal blood flow can influence GFR independently of changes in glomerular hydrostatic pressure.
With increasing renal blood flow, a lower fraction of the plasma is initially filtered out of the glomerular capillaries, causing a slower rise in the glomerular capillary colloid osmotic pressure and less inhibitory effect on GFR. Consequently, even with a constantglomerular hydrostatic pressure, a greater rate of blood flow into the glomerulus tends to increase GFR, and a lower rate of blood flow into the glomerulus tends to decrease GFR.
Increased Glomerular Capillary Hydrostatic Pressure Increases GFR
The glomerular capillary hydrostatic pressure has been estimated to be about 60 mm Hg under normal conditions. Changes in glomerular hydrostatic pres-sure serve as the primary means for physiologic regu-lation of GFR. Increases in glomerular hydrostatic pressure raise GFR, whereas decreases in glomerular hydrostatic pressure reduce GFR.
Glomerular hydrostatic pressure is determined by three variables, each of which is under physiologic control: (1) arterial pressure, (2) afferent arteriolarresistance, and (3) efferent arteriolar resistance.
Increased arterial pressure tends to raise glomeru-lar hydrostatic pressure and, therefore, to increase GFR. (However, as discussed later, this effect is buffered by autoregulatory mechanisms that maintain a relatively constant glomerular pressure as blood pressure fluctuates.)
Increased resistance of afferent arterioles reduces glomerular hydrostatic pressure and decreases GFR. Conversely, dilation of the afferent arterioles increases both glomerular hydrostatic pressure and GFR (Figure 26–14).
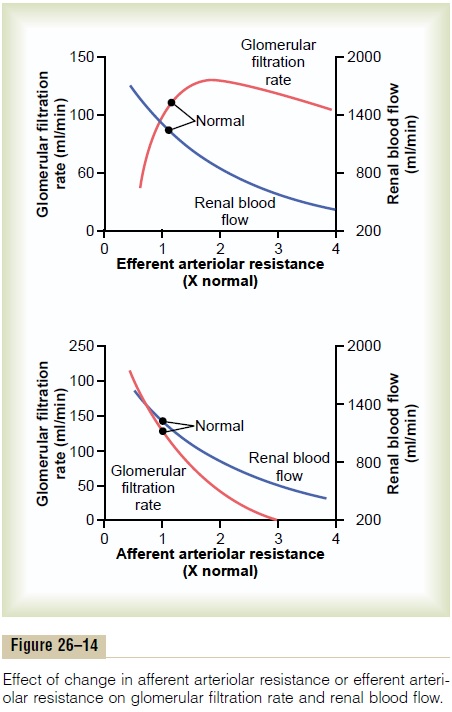
Constriction of the efferent arterioles increases the resistance to outflow from the glomerular capillaries. This raises the glomerular hydrostatic pressure, and as long as the increase in efferent resistance does not reduce renal blood flow too much, GFR increases slightly (see Figure 26–14). However, because efferent arteriolar constriction also reduces renal blood flow, the filtration fraction and glomerular colloid osmotic pressure increase as efferent arteriolar resistance increases. Therefore, if the constriction of efferent arterioles is severe (more than about a threefold increase in efferent arteriolar resistance), the rise in colloid osmotic pressure exceeds the increase in glomerular capillary hydrostatic pressure caused by efferent arteriolar constriction. When this occurs, the net force for filtration actually decreases, causing areduction in GFR.
Thus, efferent arteriolar constriction has a biphasic effect on GFR. At moderate levels of constriction, there is a slight increase in GFR, but with severe con-striction, there is a decrease in GFR. The primary cause of the eventual decrease in GFR is as follows: As efferent constriction becomes severe and as plasma protein concentration increases, there is a rapid, non-linear increase in colloid osmotic pressure caused by the Donnan effect; the higher the protein concentra-tion, the more rapidly the colloid osmotic pressure rises because of the interaction of ions bound to the plasma proteins, which also exert an osmotic effect.
To summarize, constriction of afferent arterioles reduces GFR. However, the effect of efferent arterio-lar constriction depends on the severity of the con-striction; modest efferent constriction raises GFR, but severe efferent constriction (more than a threefold increase in resistance) tends to reduce GFR.
Table 26–2 summarizes the factors that can decrease GFR.

Related Topics