Chapter: Biology of Disease: Membrane, Organelle and Cytoskeletal Disorders
Cytoskeletal Disorders
CYTOSKELETAL DISORDERS
The cytoplasm of nucleated cells is supported by a cytoskeleton
consisting of three types of fibers and a number of associated proteins. The
functions of the fibers are to resist forces that would deform the cell, to
allow the cell to change shape and move and some types of intracellular
transport. The three types of fibers are microfilaments (MF), intermediate
filaments (IF) and microtubules (MT). Although the lengths of the fibers are
indeterminate as they are actively extended and shortened during cellular
activities, their diameters are fairly uniform between cell types.
Microfilaments and MTs have diameters of approximately 7 and 25 nm
respectively. As their name implies, IFs have diameters between these values of
8–11 nm.
Microfilaments are made of the protein actin. They are
relatively flexible filaments but, cross-linked into bundles, they can
withstand compression. Microtubules are composed of tubulin proteins arranged
into hollow rods that are rigid and can resist both compression and tension.
Intermediate filaments are built up from a number of types of proteins that are
tissue specific, keratins in epidermal cells, desmin in muscles, for example.
They form flexible cables whose high tensile strength allows the cell to resist
excessive stretching.
Microfilaments and MTs form defined tracks within the cell for
the transport of macromolecules and membranous structures. The two most common
methods for this involves the movements of motor proteins along the filaments
that are driven by the hydrolysis of ATP. The motor proteins of the MTs are
dyneins and kinesins; those of the MFs are the myosins. Actin–myosin complexes
are probably best known as the contractile apparatus of skeletal muscle
tissues. Skeletal muscle tissue shows a multinuclear organization or syncitium
arranged into fibers, which are surrounded by a basal lamina of extracellular
matrix proteins, which forms a supporting sheath. Each fiber contains sarcoplasm
(cytoplasm) that houses the contractile fibers of actin and myosin and is
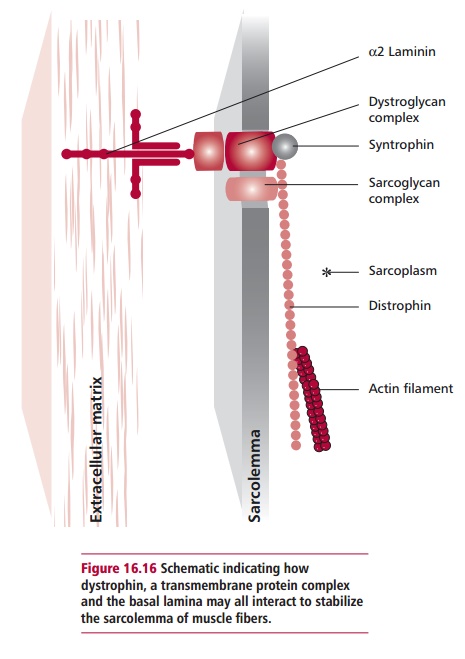
surrounded by a sarcolemma (plasma membrane). A network of elongated protein
molecules about 150 nm long of the protein dystrophin is found within the
sarcoplasm The dystrophin links actin filaments to a transmembrane complex of
proteins that, in turn, is linked to components of the basal lamina (Figure 16.16). This complex arrangement
The blood contains about 5 q 1012 erythrocytes per dm3 . Their major
function is to carry dioxygen from the lungs to the general body tissues. The
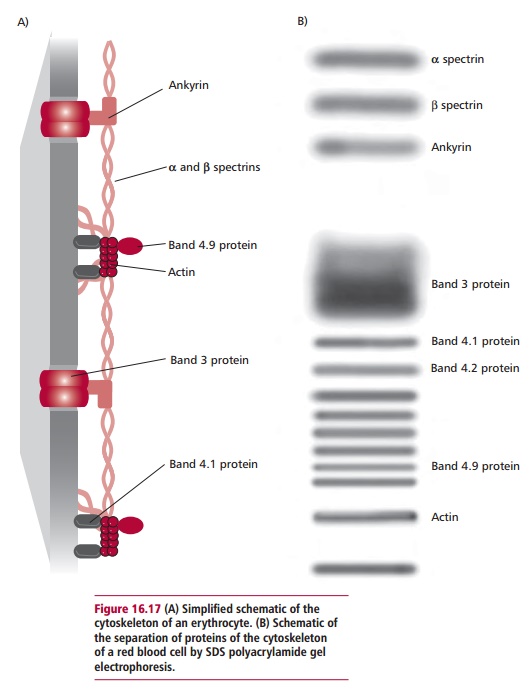
unique biconcave shape of erythrocytes is maintained by a cytoskeleton composed
of five major proteins that form a network lining the inner sur-face of their
plasma membranes (Figure 16.17). The
spectrin–actin complex is thought to act in a manner that resembles that of the
dystrophin–actin complex of skeletal muscle and provides mechanical support to
the plasma membrane preventing its lysis during circulation. The network of
proteins also allows erythrocytes to deform and spring back into shape as they
are pumped through the narrow capillaries of the vascular system. The numbers
of erythrocytes are maintained by a constant production in the bone marrow and
the destruction of worn out or misshapen erythrocytes by the spleen. This
destruction releases bilirubin, which is converted to bile salts in the liver
and released into the gastrointestinal tract in bile . The iron from the
hemoglobin is largely retained and reused by the body.
Related Topics