Introduction | Chemistry - Chemical Kinetics | 12th Chemistry : UNIT 7 : Chemical Kinetics
Chapter: 12th Chemistry : UNIT 7 : Chemical Kinetics
Chemical Kinetics
CHEMICAL
KINETICS
INTRODUCTION
We have already learnt that the feasibility of a chemical reaction under a given set of
conditions can be predicted, using the principles of thermodynamics. However,
thermodynamics does not provide an answer to a very important question of how
fast a chemical reaction takes place. We know from our practical experience
that all chemical reactions take some time for completion. Reaction speeds
ranging from extremely fast (in femto seconds) to extremely slow (in years).
For example, when the reactants BaCl2 solution and dilute H2SO4
are just mixed, a white precipitate of BaSO4 is immediately formed;
on the other hand reactions such as rusting of Iron take many years to complete.
The answers to the questions such as (i) how fast a chemical change can occur
and (ii) What happens in a chemical reaction during the period between the
initial stage and final stage are provided by the chemical kinetics. The word
kinetics is derived from the Greek word “kinesis” meaning movement.
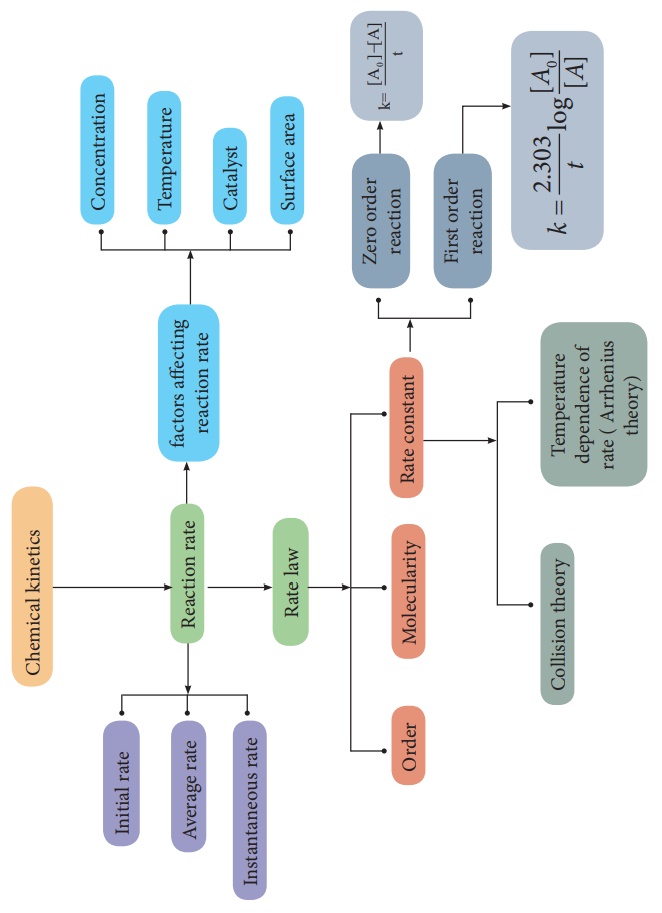
Chemical kinetics is the
study of the rate and the mechanism of chemical reactions, proceeding under
given conditions of temperature, pressure, concentration etc.
The study of chemical
kinetics not only help us to determine the rate of a chemical reaction, but
also useful in optimizing the process conditions of industrial manufacturing
processes, organic and inorganic synthesis etc.
In this unit, we discuss
the rate of a chemical reaction and the factors affecting it. We also discuss
the theories of the reaction rate and temperature dependence of a chemical
reaction.
Related Topics