Chemical Kinetics - Collision theory | 12th Chemistry : UNIT 7 : Chemical Kinetics
Chapter: 12th Chemistry : UNIT 7 : Chemical Kinetics
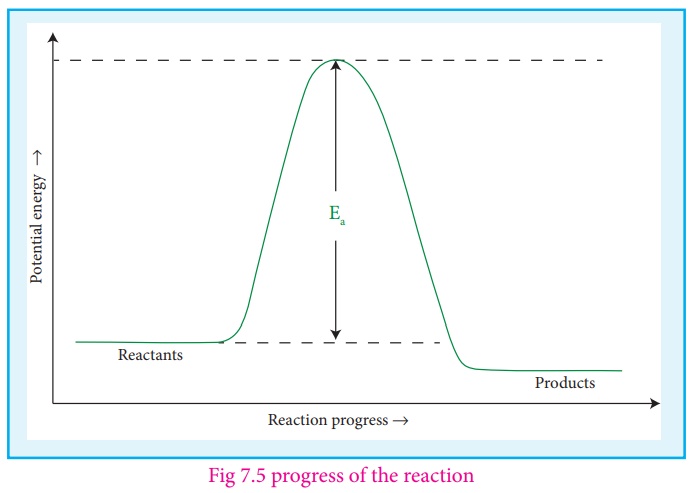
Collision theory
Collision
theory :
Collision Theory was
proposed independently by Max Trautz in 1916 and William Lewis in 1918. This
theory is based on the kinetic theory of gases. According to this theory,
chemical reactions occur as a result of collisions between the reacting
molecules. Let us understand this theory by considering the following reaction.
A 2 (g) + B2
(g) → 2AB(g)
If we consider that, the
reaction between A2 and B2 molecules proceeds through
collisions between them, then the rate would be proportional to the number of
collisions per second.
Rate ∝ number of molecules
colliding per litre per second (collision rate)
The number of collisions
is directly proportional to the concentration of both A2 and B2 .
Collision rate ∝ [A2 ][B2
]
Collision rate = Z [A2
][B2 ]
Where, Z is a constant.
The collision rate in
gases can be calculated from kinetic theory of gases. For a gas at room
temperature (298K) and 1 atm pressure, each molecule undergoes approximately 109
collisions per second, i.e., 1 collision in 10-9 second. Thus, if
every collision resulted in reaction, the reaction would be complete in 10-9
second. In actual practice this does not happen. It implies that all collisions
are not effective to lead to the reaction. In order to react, the colliding
molecules must possess a minimum energy called activationn energy. The
molecules that collide with less energy than activation energy will remain
intact and no reaction occurs.
Fraction of effective
collisions (f) is given by the following expression
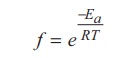
To understand the
magnitude of collision factor (f), Let us calculate the collision factor (f)
for a reaction having activation energy of 100 kJ mol−1 at 300K.
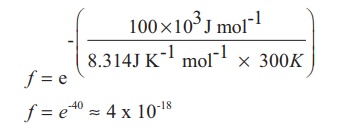
Thus, out of 1018
collisions only four collisions are sufficiently energetic to convert reactants
to products. This fraction of collisions is further reduced due to
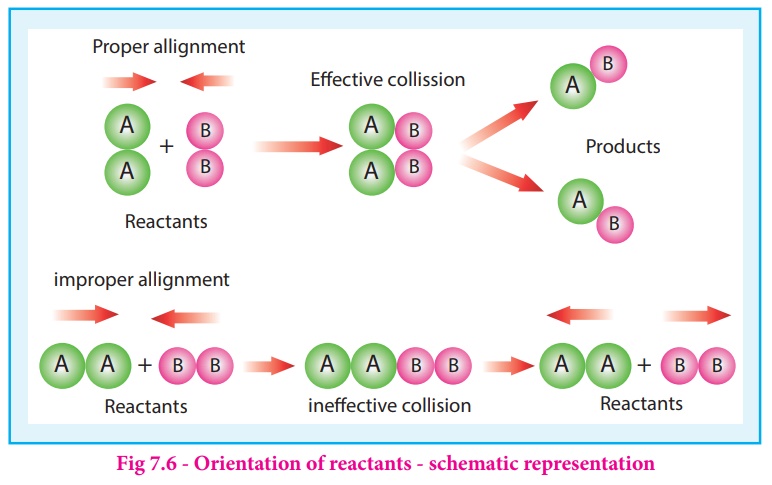
The figure 7. 6
illustrates the importance of proper alignment of molecules which leads to
reaction.
The fraction of
effective collisions (f) having proper orientation is given by the steric
factor p.
Rate = p x f x collision
rate
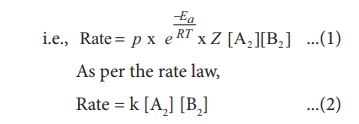
Where k is the rate constant
On comparing equation (1) and (2), the rate constant k is ‘
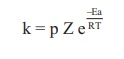
Related Topics