Chapter: 12th Chemistry : UNIT 7 : Chemical Kinetics
Arrhenius equation - The effect of temperature on reaction rate
Arrhenius
equation - The effect of temperature on reaction rate
Generally, the rate of a reaction increase with increasing temperature. However, there are very few exceptions. The magnitude of this increase in rate is different for different reactions. As a rough rule, for many reactions near room temperature, reaction rate tends to double when the temperature is increased by 10ºC .
A large number of
reactions are known which do not take place at room temperature but occur
readily at higher temperatures. Example: Reaction between H2 and O2
to form H2O takes place only when an electric spark is passed.
Arrhenius suggested that
the rates of most reactions vary with temperature in such a way that the rate
constant is directly proportional to e-(E0/RT) and he proposed a
relation between the rate constant and temperature.

Where A the frequency
factor,
R the gas constant,
Ea the activation
energy of the reaction and,
T the absolute
temperature (in K)
The frequency factor (A)
is related to the frequency of collisions (number of collisions per second)
between the reactant molecules. The factor A does not vary significantly with
temperature and hence it may be taken as a constant.
Ea is the
activation energy of the reaction, which Arrhenius considered as the minimum
energy that a molecule must have to posses to react.
Taking logarithm on both
side of the equation (1)
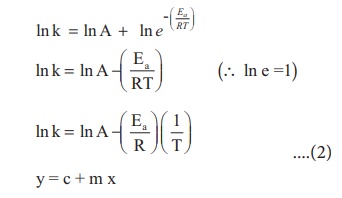
The above equation is of
the form of a straight line y = mx + c.
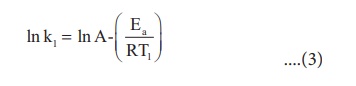
A plot of ln k Vs 1/T gives
a straight line with a negative slope – Ea/R. If the rate constant
for a reaction at two different temperatures is known, we can calculate the
activation energy as follows.
At temperature T = T1
; the rate constant k = k1
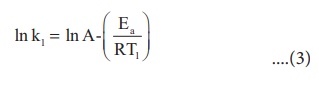
At temperature T = T2
; the rate constant k = k2
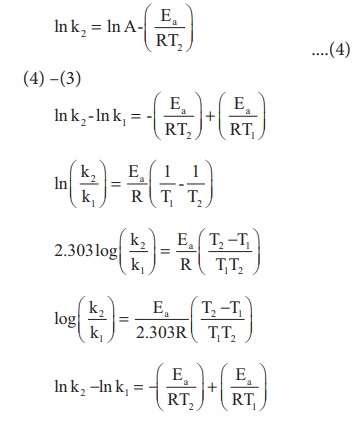
This equation can be
used to calculate from rate constants k1 and k2 at
temperatures T1 and T2 .
Example 7
The rate constant of a reaction at
400 and 200K are 0.04 and 0.02 s-1 respectively. Calculate the value of
activation energy.
Solution
According to Arrhenius equation

Ea = 2305 J mol−1
Example 8
Rate constant k of a reaction varies
with temperature T according to the following Arrhenius equation
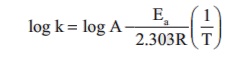
Where Ea is the activation energy.
When a graph is plotted for log k Vs 1/T a straight line with a slope of - 4000K
is obtained. Calculate the activation energy
Solution
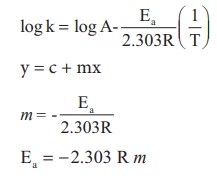
Ea = − 2.303 R m
Ea = − 2.303 x 8.314 J K−1
mol−1 x (− 4000K )
Ea = 76,589J mol−1
Ea = 76.589 kJ mol−1
Evaluate yourself
For a first order reaction the rate
constant at 500K is 8 X 10−4 s−1 . Calculate the
frequency factor, if the energy of activation for the reaction is 190 kJ mol-1
.
Related Topics