Chapter: Biology of Disease: Transfusion and Transplantation
Blood and Blood Products for Transfusion
BLOOD AND BLOOD PRODUCTS FOR
TRANSFUSION
The role of the biomedical scientist in the
transfusion laboratory is to ensure that the blood and blood products being
transfused into a patient are safe. To ensure safety, the blood is tested to
determine its blood group and to check that it is not contaminated with harmful
microorganisms. In addition, checks are made to ensure that the transfused
blood does not contain anti-bodies that will destroy the erythrocytes of the
recipient and cause death.
Blood transfusions are required to replace blood lost
as a result of accident or surgery. Surgical procedures which require
transfusions include the trans-plantation of organs, such as the liver and
heart, where significant bleeding may occur. Blood may also be given to treat
certain diseases, such as anemia. Plasma may also be transfused to treat badly
burned patients who have lost significant amounts of fluid or in the treatment
of bleeding disorders. Plasma products, such as Factor VIII, to treat
hemophilia , or immu-noglobulins, to treat certain immunodeficiency disorders ,
may also be given. Platelet concentrates are also used to treat bleeding
disorders (Figure 6.1).
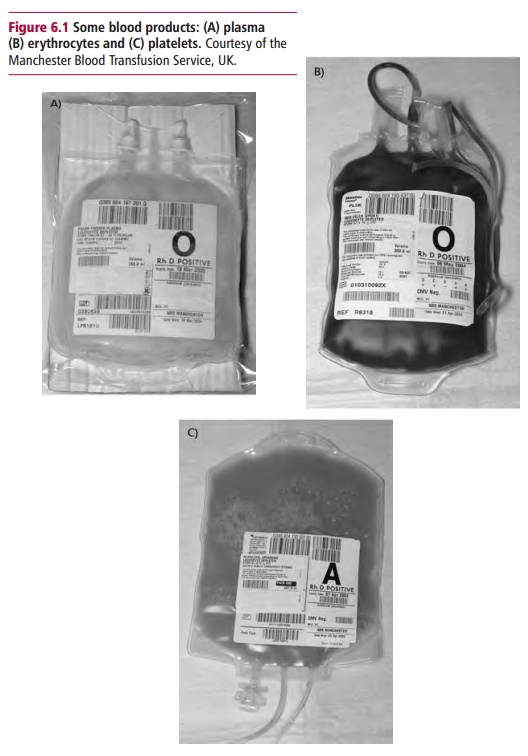
In the UK, blood containing leukocytes is no longer
transfused for a number of reasons, as shown in Table 6.1. Sensitization to Major Histocompatibility Complex (MHC)
antigens, which are present on blood leukocytes but not erythrocytes, may have
consequences if the recipient later requires a trans-plant and GVHD may have a fatal outcome in
immunosup-pressed individuals. As a consequence, leukocytes are removed from
blood, usually within a few hours of collection. This involves filtering the
blood through leukocyte-specific filters, which trap the leukocytes but not the
smaller erythrocytes or platelets. Such a process is called leukodepletionand it reduces the
leukocyte count to less than 5 q 106 dm –3. The number of
leu-kocytes left in blood can be assessed by counting in a hemocytometer, or by
using a flow cytometer.
Related Topics