Chapter: Aquaculture Principles and Practices: Health and Diseases
Bacterial diseases of aquaculture species
Bacterial diseases
There are several bacterial diseases of cultured fish and shellfish and, as mentioned earlier, many bacterial infections occur in association with viral diseases, as secondary infections. Because of the association of different pathogens, the identity of diseases and description of causative organisms in the literature may appear confusing. Bacterial diseases have worldwide distribution and occur in both tropical and temperate climate aquacultures. While sanitation and prevention are the measures of choice to control the disease, there are proven chemotherapeutic agents that can be successfully used in treating many bacterial diseases when they occur.
Furunculosis
Furunculosis is a septicaemic bacterial disease occurring mainly in salmonids. The disease has almost worldwide distribution and also infects many species of cold-water and warm-water fishes, besides salmonids. The name furunculosis is derived from the furuncules or blisters that occur on the skin of infected fish (fig. 9.7). But this is not a sure sign of the disease as furuncules can occur in other types of infections as well and also because, in acute cases of furunculosis, furuncules may not be present. The gram-negative bacterium Aeromonassalmonicida, which is the causative agent, may
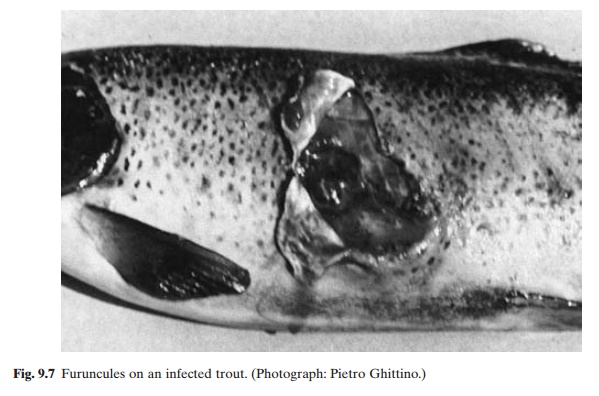
survive for days or weeks in water, but cannot persist indefinitely in the absence of a carrier fish. The classic salmonid furunculosis is caused by strains of A. salmonicidawhich produce brown pigment on bacteriological media. The ulcer disease in goldfish, the carp erythroder-matitis and certain disease conditions in some other warm-water fishes are consequences of infections with strains or subspecies of A.salmonicida which do not produce such brownpigment.
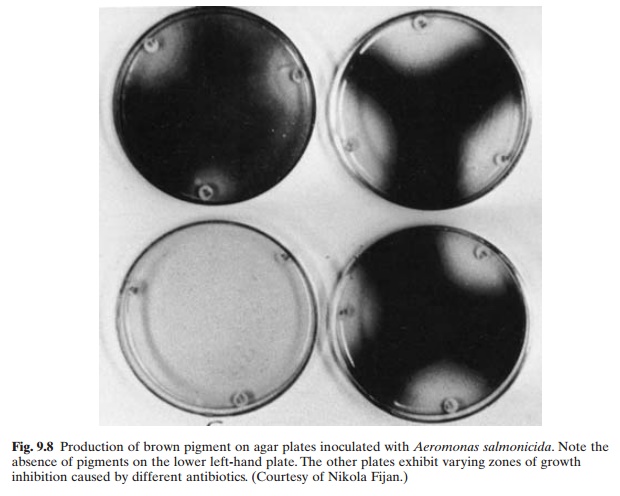
Positive diagnosis has to be based on isolation and identification of the causative agent. The organism is typically a gram-negative, non-motile rod that ferments selected carbohydrates, produces cytochrome oxidase and yields a water-soluble brown pigment on several types of isolation agar (fig. 9.8). But special care is recommended in identifications as a number of atypical achromogenic variants have been reported. Asymptomatic carriers are very difficult to detect. To establish the absence of carriers in a brood stock, the use of serum agglutination techniques or corticosteroid techniques might have to be employed (Bullock and Stuckey, 1975).
The incubation period for acute cases is probably two to four days, but in chronic cases the period may be extended by several weeks at lower temperatures. Furunculosis is usually seasonal, with peak incidence during the mid-summer months of July and August.
In carp erythrodermatitis and goldfish ulcer disease, clinical signs appear first only on the skin: small local erosions surrounded by an inflamed reddish zone developed gradually into large and sometimes deep ulcers. Seriously affected moribund fishes also have protruding eyes, fluid in the body cavity and oedematous organs. The causal agent can be isolated from the inflamed skin. Fish which recover from the disease can have scars on the skin.
Epizootics can be treated with medicated feeds. Terramycin (oxytetracycline), added to feed at the rate of 3 g per 45 kg fish, can be administered daily for 10 days. The recom
mended dosage of sulphamerazine is 5–10 g for 45 kg fish, fed for 10–15 consecutive days.
Sulphonamides or nitrofurans can also be used, as well as antibiotics. It should be remembered, however, that in some areas the stocks have developed resistance to terramycin and sulphamerazine, probably due to the feeding of low levels of these antibiotics as a prophylactic measure.
Columnaris disease
Columnaris disease occurs in acute or chronic form in both cold-water and warm-water fishes worldwide, and is caused by the bacterium Flexibacter columnaris (fig. 9.9). Strains of high andlow virulence have been identified. The highly virulent form attacks the gill tissue and the less virulent ones are primarily responsible for cutaneous infections.
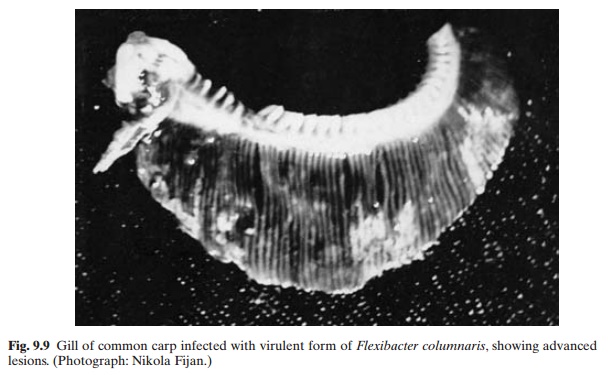
The first sign of the disease may be the appearance of discoloured grey patches in the dorsal fin area. These lesions grow and expose the underlying muscle tissue. The lesions are prominent in the mouth and head regions and may become yellow and cratered. The infection of virulent strains cause a ‘gill rot’ condition, whereas the systemic infections by less virulent strains may not show any external signs. Cutaneous infections are prevalent among most species of fish.
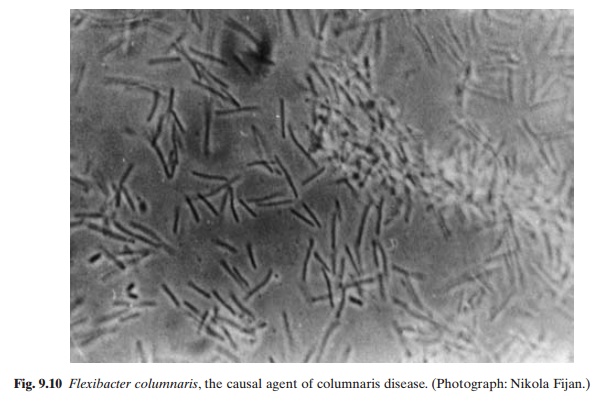
A preliminary diagnosis of the disease can be made based on the detection of long, slender, gram-negative rods in smears of gills or scrappings from cutaneous lesions (fig. 9.10). The diagnosis can be confirmed by isolation of the organism on cytophaga medium. Colonies of F.columnaris exhibit a rough, rhizoid-marginatedgrowth that tends to extend into the agar.
Infected fish with gill or cutaneous lesions serve as a source of infection; fish under stress due to elevated temperatures and crowding are susceptible to infection. The period between exposure and outbreak of the clinical disease varies, depending on the virulence of the bacterial strain and the ambient water temperature. High-virulence strains may induce acute
disease within 24 hours, whereas less virulent forms may require from 48 hours to several weeks. The disease definitely has a pronounced seasonal occurrence, and is concentrated during the warm summer months.
Avoidance of exposure to the disease, use of disinfected water supplies, reduced population density and maintenance of lower temperature (in the case of cold-water species below 13°C) are possible means of preventing the disease.
Prophylactic treatments can be adopted when other measures are not feasible. Baths of copper sulphate (CuSO4) for 20 minutes at 33 ppm or of potassium permanganate (KMnO4) at 2 ppm for indefinite periods have been recommended; but care should be taken in their administration, since these chemicals can be toxic to certain species in soft waters. Such baths have also been recommended in chemotherapy of the disease. Besides CuSO4 and KMnO4, quaternary ammonium compounds at doses of 2–3 ppm in one-hour flow-through treatments have been reported to be effective for salmonids. Oxytetracycline (terramycin) incorporated into the food at the rate of 4 g per 45 g fish, fed at 3 per cent body weight per day, is the usual antibiotic treatment
Bacterial gill disease (BGD)
Bacterial gill disease is an external infection of hatchery-reared salmonids and occasionally of intensively reared warm-water species. It appears to be caused by one or more species of filamentous bacteria, including Flavobacterium sp. Large numbers of filamentous bacteria can be observed on the gills, accompanied by fusing and clubbing of the gill filaments. The disease generally occurs following the deterioration of environmental conditions, associated with over-crowding and accumulation of toxic metabolic products. Acute or chronic forms of the disease may occur.
Infected fish are usually lethargic and apparently lose appetite. Acute epizootics may result in high mortality of up to 50 per cent in a day. Although extensive clubbing of the gill filaments, lamellar fusion and excess mucus may be found, necrosis of the gill tissue seldom occurs. This is in contrast to columnaris disease, whereextensive necrosis and erosion of the gill filaments can be observed.
The detection of large numbers of filamentous bacteria on the gills under a microscope (under a wet mount or stained with methylene blue) is the recommended diagnostic procedure. Isolation of a pure culture is not considered necessary.
The biology and survival of the aetiological organism are not fully known. Contaminated water or carrier fish are probably the source of infection, but it is almost always associated with deterioration of environmental conditions. Fingerlings are generally more susceptible to the infection and salmonids over one year in age seldom develop the disease.
Application of proper sanitation practices, avoidance of crowding and reuse of water, and maintenance of an adequate flow of clean water should help to reduce the incidence of the disease. A number of compounds have been found to be effective for treatment of BGD, but most of them require multiple applications. Successful treatments are (i) potassium permanganate (KMnO4) at 1–2 ppm, (ii) Hyamine
1622 and 3500, as well as Roccal at 1–2 ppm calculated on the basis of active ingredient, (iii) Diquat at 8.4–16.8 ppm of the formulation, (iv) another quaternary ammonium compound Purina Four Power, at 3–4 ppm as a one-hour flush treatment, and (v) chloramine-T at 10 ppm, in a single one-hour flush treatment.
Gill rot
Gill rot disease is reported to be one of the main infectious diseases in China and affects especially grass carp (Ctenopharyngodonidella) fingerlings. The causative organism hasbeen described as Myxococcus piscicola. Whether it is the same as the bacterial gill disease described as affecting juvenile salmonids (Snieszko, 1958) is not clear. In juvenile salmonids, it can occur over a wide temperature range and is characterized by masses of myxobacteria on the gills and associated with hyperplasia of the gill epithelium. Several kinds of myxobacteria can be isolated from the gills, though not from the internal organs. It is suspected that the disease is secondary to some other predisposing stress factor and that the myxobacteria are only opportunists. Chinese workers have isolated Myxococcus piscicola from the affected gills. The disease symptoms are described as pale coloration of the gill filaments, then sloughing and accumulation of excess slime. The gill covers of seriously infected fish are inflamed and are eroded by the bacteria forming small transparent patches. The disease is reported to be prevalent in ponds overloaded with organic matter.
Farmers are advised to treat the infected ponds with bleaching powder (containing about 30 per cent available chlorine), at the rate of about 4 kg/ha. In serious cases, a treatment dose of 1 ppm is recommended. For salmonid gill disease, the addition of anti-bacterial chemicals to the water is suggested, as the infection is restricted to the gills. Hyamine 1622 at 2 ppm of commercial product or Diquat at 2 or 4 ppm of active ingredient can be used as one-hour baths daily for two to four days.
Enteric red mouth disease (ERM)
Enteric red-mouth disease is an acute to chronic bacterial disease of intensively cultured
rainbow trout and other salmonids. The causative agent is a motile, gram-negative, rod-shaped bacterium, identified as Yersinia ruckeri, transmitted from fish to fish by contact and through water. It causes sustained low-level losses, but in severe epizootics mortality rates can be higher, exceeding 50 per cent. Surviving fish frequently become asymptomatic carriers.
Acute cases are seldom detected. The clinical symptoms are similar to infections of
Aeromonas hydrophila and A. salmonicida. Theaffected fish become dark and lethargic and refrain from feeding. Surviving carrier fish also show the same signs and may often have missing eyes or exophthalmia, with little avoidance reaction. During the acute stage of the disease, small bright haemorrhages occur along the gumline of the mouth and on the tongue, which together with the general inflammation give the characteristic ‘red mouth’ appearance (fig. 9.11). Small haemorrhages may develop on the belly and also at the base of the fins. A flaccid, fluid-filled stomach and haemorrhages of the mouth, when occurring together, give positive evidence of the disease. An enlarged dark spleen, haemorrhagic specks on the air bladder and pyloric caecae, and reddening of the posterior intestinal tract are other internal signs of the infection. Confirmatory diagnosis consists of the isolation of gram-negative motile rods of Y. ruckeri, with positive agglutination with rabbit anti-Y. ruckeri serum. The source of infection of the disease is asymptomatic carriers. No evidence of transmission from parent to progeny through eggs has been reported.
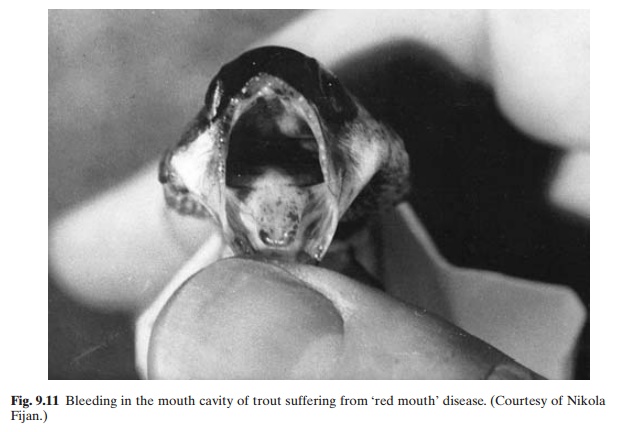
Susceptibility to the disease is obviously related to stress caused by handling or culture conditions. There does not seem to be any seasonality in its occurrence. The incubation period in young rainbow trout (7.5–10 cm) appears to be 5–19 days at about 15°C, and in young Atlantic salmon (6 cm) nine days after exposure at 12.5°C.
As detection of the pathogen in apparently healthy carrier fish is difficult, regular health inspection and monitoring are essential to prevent the disease. Disinfection of water supplies to hatcheries and rearing facilities is another means of disease prevention. Commercial vaccines against this disease are available, and though they may not always provide total protection, they can help to control the infection and reduce losses. Therapeutics recommended are sulphamerazine and oxytetracycline (terramycin). Vaccination and/or chemotherapy should be accompanied by avoidance of adverse environmental factors and excessive handling stresses.
Edwardsiellosis
Edwardsiellosis, caused by infection by Edwardsiella tarda, affects Ictalurids, Cyprinidsand Anguillidae in the southern USA and Southeast Asia. It causes gas-filled lesions in the muscle tissues of mature fish. In the initial stages of the infection and in mild infections, small cutaneous lesions (of 3–5 mm diameter) can be observed on the flanks (fig. 9.12) and caudal peduncle of the fish. Mortality seldom exceeds 5 per cent in affected channel catfish in ponds, but may reach 50 per cent or more if the fish are transferred to holding tanks.
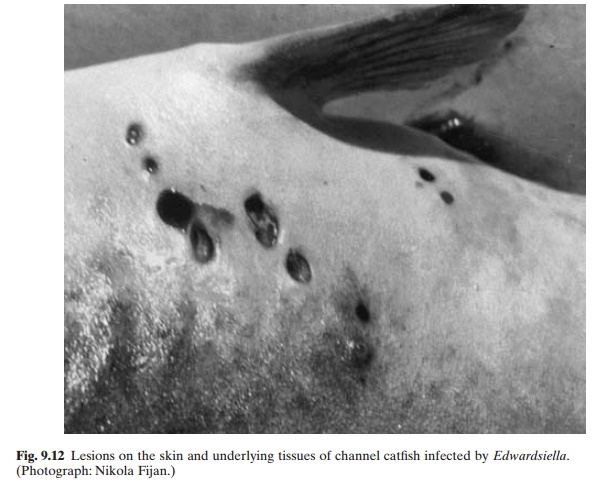
Colonies of E. tarda are dirty grey in colour and smooth. The organism is motile, gram-negative and fermentative. It occurs in organically polluted waters. Lesions develop in the infected fish 10–15 days after exposure, becoming large gas-filled cavities containing necrotic tissue. Bacteria can frequently be detected in the blood or kidney of the fish and evidently cause disfunction of the organs. Infection by E. tarda may often occur in association with Aeromonas or Pseudomonas infections and therefore confusion can arise in diagnosis. Recovery from the disease is rather slow. Prevention of the disease has to be attempted through avoidance of environmental stresses, improved husbandry practices and better nutrition. Treatment with antibacterials is reported to be effective. Oxyte-tracycline (terramycin) in the diet at the rate of 2.5 g per 45 kg fish per day for 10–12 days is the recommended dosage. Sulphonamide or furacin have also been reported to be effective.
Vibriosis
Vibriosis, caused by the bacterium Vibrioanguillarum which occurs in both fresh- andsalt-water, has become one of the most serious diseases of cultivated marine species of fish and
Norway. Rainbow trout, pink salmon and char can be attacked at any size or age, but Atlantic salmon are normally vulnerable only in the parr and smolt stages of development, and perhaps as they approach maturity. Sea-pen culture of Pacific salmon, particularly of coho salmon, was threatened by vibriosis until methods of control by antibiotics and immunization were developed.
Vibriosis in salmon normally occurs in smolt units, which pump water from the sea, and in sea units soon after the smolts are stocked. The vibrio usually enters the fish through surface wounds and acts mostly on the skin, where lesions are formed. The ulcers can extend deep into the muscles and internal haemorrhage, kidney damage and a swollen spleen are sometimes found in dying fish. Vibriosis is essentially a disease of spring and summer and the growth of the bacterium appears to be accelerated at higher temperatures. The bacterial septicaemia in shrimps appears to be mainly caused by vibrios, even though Pseudomonas sp. and Aeromonas sp. are generally associated withthe disease. Disoriented swimming and increasing opaqueness of abdominal muscle in juve-niles and adults are common signs of the infection. Infection occurs in larvae, juveniles and adult shrimps. Complete mortality of the stock can occur.
Recommended preventive measures for salmonids are reduction of stocking density in warm waters and disinfection of eggs brought into the hatchery. Immunization by injection of juveniles or by hyperosmotic spray and bath immunization has been found to be useful. Oral immunization has been effective in some experiments, but is not recommended for use in commercial aquaculture. The appearance of new strains or serotypes of V. anguillarum has made it necessary to incorporate them also into the immunization protocol. Some cross-protection is provided by various vaccines but not enough, and not consistently (Sindermann, 1986).
Treatment of vibrios is by addition of drugs to the food, but since in the advanced stages of infection the fish stops feeding, treatment has to be started early. The suggested dosages are 0.2 g of sulphamerazine per kg fish per day for three days first of all, then after an interval of a day or two another treatment of 0.2 g per kg fish for a further three days. An alternative treatment is oxytetracycline or furazolidone at the rate of 50–75 mg per kg fish for up to 10 days. The suggested treatment for shrimps is oxytetracycline at the rate of 40 mg per kg body weight per day with feed or 1 ppm baths for the larval stages (zoeae and mysis) every 48 hours. Alternatively, furanace can be used at the rate of 100 mg per feed or 1 ppm baths for the larval stages every 48 hours.
The brown spot disease of shrimps also appears to be primarily caused by vibrios, but as in the case of bacterial septicemia, other organisms like Pseudomonas sp. andBeneckea are implicated. The infection affects adult and juvenile shrimps. Brownish, eroded areas can be seen on the exoskeleton. The lesion generally results from a break on the exoskeleton, as a result of physical stress. The eroded areas may become portals of entry for secondary pathogens, causing mortality. The infection may be eliminated at moulting, except when under-lying tissues are affected. Suggested treatment is administration of oxytetracycline at the rate of 450 mg per kg feed, or external treatment with 0.05–1 ppm malachite green or 20–75 ppm formalin.
Other bacterial diseases
Bacterial infections by the genus Pseudomonas are common in cold- and warm-water fishes.
Identification of the responsible species can be done by serological methods. P. fuorescens infections in catfish have been controlled either by intraperitoneal injections of kanamycin (25 mg per 0.45 kg body weight) or by feeding terramycin in the daily ration (2.5 g per 45 kg fish per day). Infected trout have been successfully treated with chloramphenicol and terramycin.
There are a number of other poorly under-stood diseases in which bacterial infection is implicated. Although bacteria have been isolated in such cases, the actual role of the bacteria has not been determined.These include such diseases known as fin rot, fin and tail rot and peduncle disease.
The name fin rot or fin and tail rot has been given to a condition in which the margins of the fins become necrotic and slough away.
Generally, the condition is associated with unfavourable environmental conditions. Under pond conditions, improvement of water quality will often result in cure. Low levels of terramycin, aureomycin and streptomycin have been found to be effective. A prophylactic treatment of potassium permanganate or 3 ppm acriflavin can also be most useful.
Pasteurella infections are sometimes found in marine species of fish, including the yellow-tail, Seriola quinqueradiata. The symptoms of the disease are very similar to haemorrhagic septicaemia and are caused by motile gram-negative rods, showing bipolarity, especially when stained with methylene blue. The bacteria are readily isolated from the kidney, spleen and liver of affected fish. Suggested treatment is by the use of antibiotics, administered through feed.
The filamentous bacterial disease of shrimps is caused by Leucothrix sp. and affects larvae and post-larvae. It is generally caused by poor water quality. Growth of filamentous bacteria on the body surface and gill tips is characteristic of the disease. Heavy infestations inhibit normal swimming and cause asphyxiation. The recommended treatment is the use of Cutrine Plus at 0.15 ppm for 24 hours in a flow-through bath or 0.5 ppm for four to six hours in a static bath.
Related Topics