Chapter: Basic & Clinical Pharmacology : Antiviral Agents
Antiviral Agents
Antiviral Agents
Viruses
are obligate intracellular parasites; their replication depends primarily on
synthetic processes of the host cell. Therefore, to be effective, antiviral
agents must either block viral entry into or exit from the cell or be active
inside the host cell. As a corollary, non-selective inhibitors of virus
replication may interfere with host cell function and result in toxicity.
Progress
in antiviral chemotherapy began in the early 1950s, when the search for anticancer
drugs generated several new compounds capable of inhibiting viral DNA
synthesis. The two first-generation antiviral agents, 5-iododeoxyuridine and
trifluorothymidine, had poor specificity (ie, they inhibited host cell DNA as
well as viral DNA) that rendered them too toxic for systemic use. However, both
agents are effective when used topically for the treatment of herpes keratitis.
Knowledge
of the mechanisms of viral replication has provided insights into critical
steps in the viral life cycle that can serve as potential targets for antiviral
therapy. Recent research has focused on identifying agents with greater
selectivity, higher potency, in vivo stability, and reduced toxicity. Antiviral
therapy is now avail-able for herpesviruses, hepatitis C virus (HCV), hepatitis
B virus (HBV), papillomavirus, influenza, and human immunodeficiency virus
(HIV). Antiviral drugs share the common property of being virustatic; they are
active only against replicating viruses and do not affect latent virus. Whereas
some infections require monotherapy for brief periods of time (eg, acyclovir
for herpes simplex virus), others require dual therapy for prolonged periods of
time (inter-feron alfa/ribavirin for HCV), whereas still others require
multiple drug therapy for indefinite periods (HIV). In chronic illnesses such as viral hepatitis and
HIV infection, potent inhibition of viral rep-lication is crucial in limiting
the extent of systemic damage.
ACRONYMS & OTHER NAMES
3TC Lamivudine
AZT Zidovudine
(previously azidothymidine)
CMV Cytomegalovirus
CYP Cytochrome P450
d4T Stavudine
ddC Zalcitabine
ddI Didanosine
EBV Epstein-Barr
virus
FTC Emtricitabine
HBeAg Hepatitis e
antigen
HBV Hepatitis B
virus
HCV Hepatitis C
virus
HHV-6 Human
herpesvirus-6
HIV Human immunodeficiency
virus
HSV Herpes simplex
virus
NNRTI Nonnucleoside
reverse transcriptase inhibitor
NRTI Nucleoside/nucleotide
reverse transcriptase inhibitor
PI Protease
inhibitor
RSV Respiratory
syncytial virus
SVR Sustained viral
response
UGT1A1 UDP-glucuronosyl transferase 1A1
VZV Varicella-zoster
virus
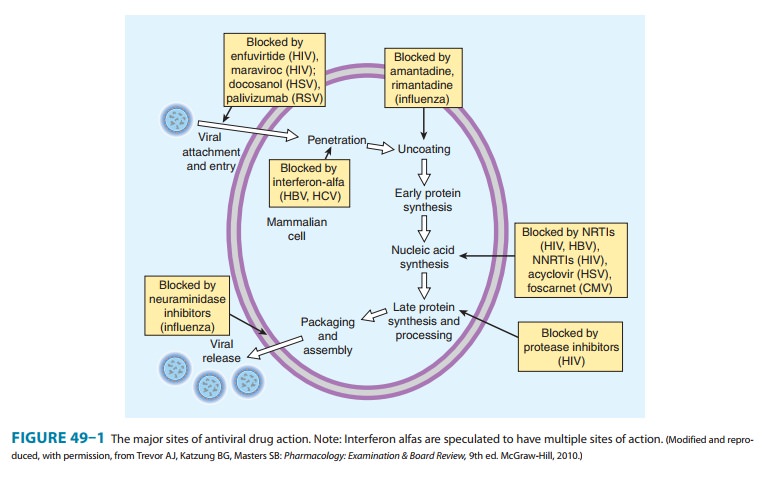
Viral
replication requires several steps (Figure 49–1): (1) attach-ment of the virus
to receptors on the host cell surface; (2) entry of the virus through the host
cell membrane; (3) uncoating of viral nucleic acid; (4) synthesis of early
regulatory proteins, eg, nucleic acid polymerases; (5) synthesis of new viral
RNA or DNA; (6) syn-thesis of late, structural proteins; (7) assembly
(maturation) of viral particles; and (8) release from the cell. Antiviral
agents can poten-tially target any of these steps.
CASE STUDY
A 35-year-old white woman who recently tested seropositive for both HIV and hepatitis B virus surface antigen is referred for evaluation. She is feeling well overall but reports a 25-pack-year smoking history. She drinks 3–4 beers per week and has no known medication allergies. She has a history of heroin use and is currently receiving methadone. Physical examination reveals normal vital signs and no abnormalities. White blood cell count is 5800 cells/mm 3 with a normal differential, hemoglobin is 11.8 g/dL, all liver function tests are within normal limits, CD4 cell count is 278 cells/mm3, and viral load (HIV RNA) is 110,000 copies/ mL. What other laboratory tests should be ordered? Which antiretroviral medications would you begin?
CASE STUDY ANSWER
Combination antiviral
therapy against both HIV and hepatitis B virus (HBV) is indicated in this
patient, given the high viral load and low CD4 cell count. However, the use of
methadone and possibly excessive alcohol consumption necessitate cau-tion.
Tenofovir and emtricitabine (two nucleoside/nucleotide reverse transcriptase
inhibitors) would be a potentially excel-lent choice as components of an
initial regimen, since both are active against HIV-1 and HBV, do not interact
with methadone, and are available in a once-daily, fixed-dose combination.
Efavirenz, a nonnucleoside reverse tran-scriptase inhibitor, could be added and
still maintain a once-daily regimen. Prior to initiation of this regimen, renal
function should be checked, and a bone mineral density test should be
considered. Pregnancy should be ruled out, and the patient should be counseled
that efavirenz should not be taken during pregnancy. The potential for lowered
metha-done levels with efavirenz necessitates close monitoring and possibly an
increased dose of methadone.
Related Topics