Chapter: Basic & Clinical Pharmacology : Antiviral Agents
Antiretroviral Agents
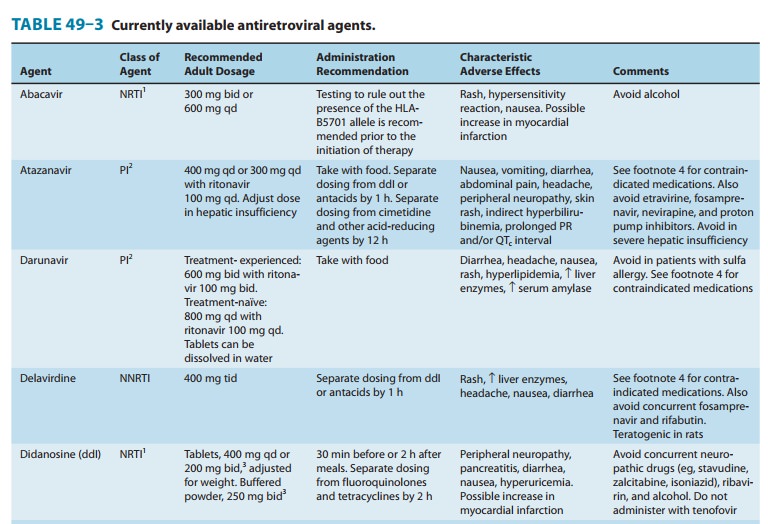
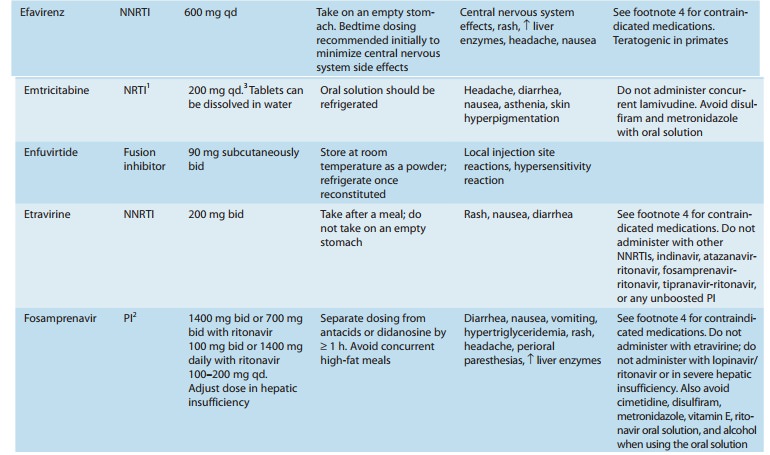
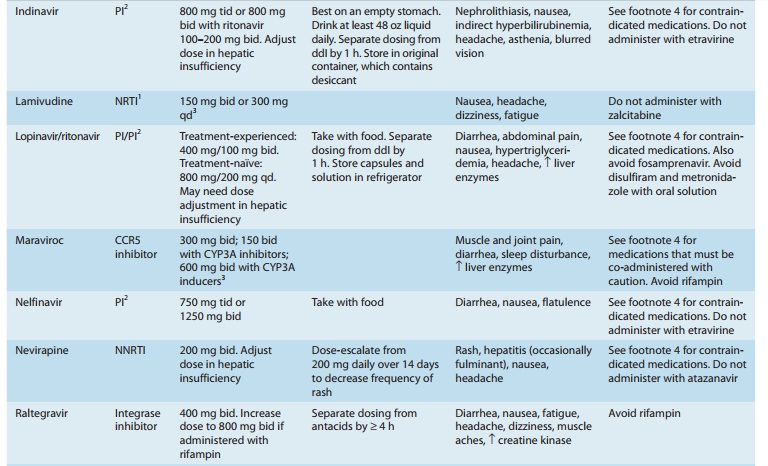
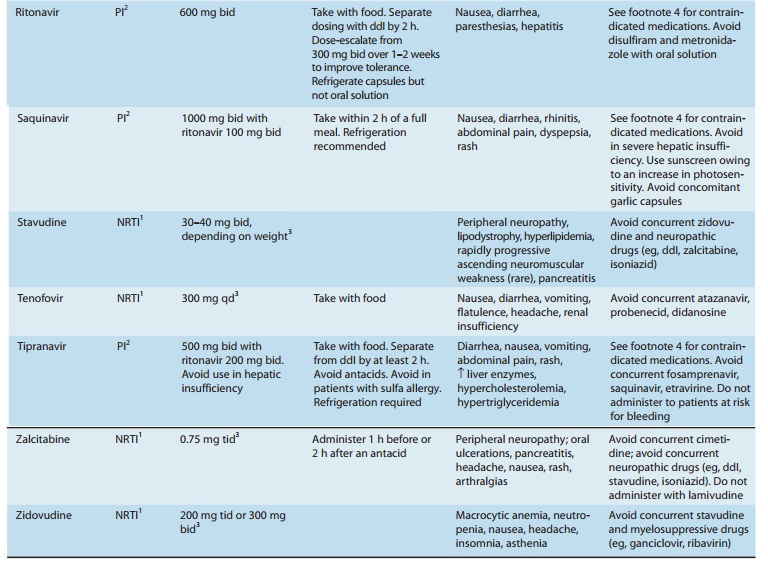
ANTIRETROVIRAL AGENTS
Substantial advances
have been made in antiretroviral therapy since the introduction of the first
agent, zidovudine, in 1987 (Table 49–3). Greater knowledge of viral dynamics
through the use of viral load and resistance testing has made it clear that
combina-tion therapy with maximally potent agents will reduce viral
replica-tion to the lowest possible level and decrease the likelihood of
emergence of resistance. Thus, administration of combination antiretroviral
therapy, typically comprising at least three antiretro-viral agents, has become
the standard of care. Viral susceptibility to specific agents varies among
patients and may change with time, owing to development of resistance.
Therefore, such combinations must be chosen with care and tailored to the
individual, as must changes to a given regimen. In addition to potency and
susceptibil-ity, important factors in the selection of agents for any given
patient are tolerability, convenience, and optimization of adherence.
The retroviral genomic
RNA serves as the template for synthe-sis of a double-stranded DNA copy, the
provirus (Figure 49–4). Synthesis of the provirus is mediated by a
virus-encoded RNA-dependent DNA polymerase, or “reverse transcriptase.” The
provirus is translocated to the nucleus and integrated into host DNA.
Transcription of this integrated DNA is regulated primarily by cellular
machinery.
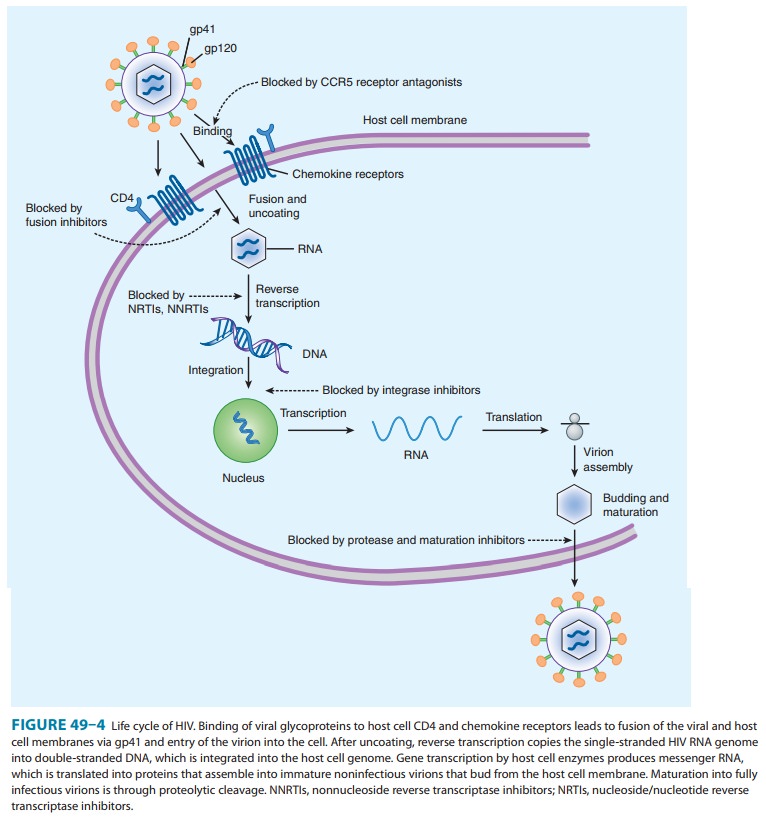
Six classes of
antiretroviral agents are currently available for use: nucleoside/nucleotide
reverse transcriptase inhibitors (NRTIs), nonnucleoside reverse transcriptase
inhibitors (NNRTIs), protease inhibitors (PIs), fusion inhibitors, CCR5
receptor antagonists, and integrase inhibitors. As new agents have become
available, several older ones have had diminished usage, because of either
suboptimal safety profile or inferior antiviral potency. It is important to
recog-nize that the high rate of mutation of HIV-1 per replication cycle
results in a great potential for genotypic variation. Genotypic resis-tance has
been reported for each of the antiretroviral agents cur-rently in use.
Treatment that slows or stops replication is critical in reducing the number of
cumulative mutations, as is the use of combinations of agents with differing
susceptibility patterns.
It should be noted that in vitro susceptibility of HIV-2 to the NRTIs is similar to that of HIV-1, albeit with a lower genetic barrier to resistance. There is innate resistance of HIV-2 to the NNRTIs, due to a different structure of the reverse transcriptases’ NNRTI binding pockets; enfuvirtide has no activity against HIV-2. Data on the activity of PI agents and maraviroc against HIV-2 are sparse and inconclusive.
Related Topics