Chapter: Basic & Clinical Pharmacology : Drugs Used in the Treatment of Gastrointestinal Diseases
Antiemetic Agents
ANTIEMETIC AGENTS
Nausea and vomiting
may be manifestations of a wide variety of conditions, including adverse
effects from medications; systemic disorders or infections; pregnancy;
vestibular dysfunction; central nervous system infection or increased pressure;
peritonitis; hepa-tobiliary disorders; radiation or chemotherapy; and
gastrointesti-nal obstruction, dysmotility, or infections.
PATHOPHYSIOLOGY
The brainstem
“vomiting center” is a loosely organized neuronal region within the lateral
medullary reticular formation and coor-dinates the complex act of vomiting
through interactions with cranial nerves VIII and X and neural networks in the
nucleus tractus solitarius that control respiratory, salivatory, and vasomo-tor
centers. High concentrations of muscarinic M1, histamine H1, neurokinin 1 (NK1), and serotonin 5-HT3 receptors have been
identified in the vomiting center (Figure 62–6).
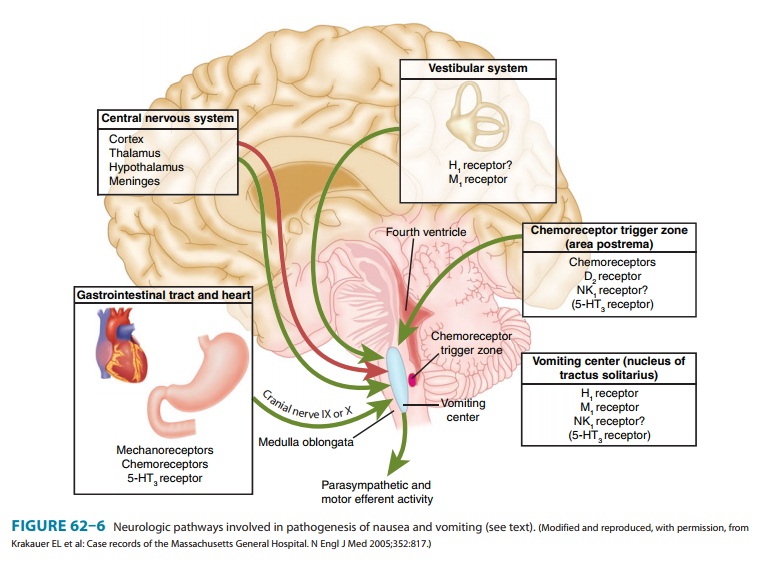
There are four
important sources of afferent input to the vomiting center:
1.
The “chemoreceptor trigger zone” or area postrema is located at the caudal end
of the fourth ventricle. This is outside the blood-brain barrier but is
accessible to emetogenic stimuli in the blood or cerebrospinal fluid. The
chemoreceptor trigger zone is rich in dopamine D2
receptors and opioid receptors, and possibly serotonin 5-HT3
receptors and NK1 receptors.
2.
The vestibular system is important in motion sickness via
cra-nial nerve VIII. It is rich in muscarinic M1 and histamine H1 receptors.
3. Vagal and spinal
afferent nerves from the gastrointestinal tract are rich in 5-HT3 receptors. Irritation
of the gastrointestinal mucosa by chemotherapy, radiation therapy, distention,
or acute infectious gastroenteritis leads to release of mucosal sero-tonin and
activation of these receptors, which stimulate vagal afferent input to the
vomiting center and chemoreceptor trig-ger zone.
4.
The central nervous system plays a role in vomiting due to
psychiatric disorders, stress, and anticipatory vomiting prior to cancer
chemotherapy.
Identification of the
different neurotransmitters involved with emesis has allowed development of a
diverse group of antiemetic agents that have affinity for various receptors.
Combinations ofantiemetic agents with different mechanisms of action are often
used, especially in patients with vomiting due to chemotherapeu-tic agents.
SEROTONIN 5-HT3 ANTAGONISTS
Pharmacokinetics & Pharmacodynamics
Selective 5-HT3-receptor antagonists
have potent antiemetic properties that are mediated in part through central
5-HT3-receptor blockade in
the vomiting center and chemoreceptor trigger zone but mainly through blockade
of peripheral 5-HT3 receptors on extrinsic intestinal vagal and spinal afferent
nerves. The anti-emetic action of these agents is restricted to emesis
attributable to vagal stimulation (eg, postoperative) and chemotherapy; other
emetic stimuli such as motion sickness are poorly controlled.
Four agents are
available in the USA: ondansetron, granise-tron,
dolasetron, and palonosetron. (Tropisetron is anotheragent available
outside the USA.) The first three agents (ondanse-tron, granisetron, and
dolasetron, Figure 62–5) have a serum half-life of 4–9
hours and may be administered once daily by oral or intravenous routes. All
three drugs have comparable efficacy and tolerability when administered at
equipotent doses. Palonosetron is a newer intravenous agent that has greater
affinity for the 5-HT3 receptor and a long serum half-life of 40 hours. All four
drugs undergo extensive hepatic metabolism and are eliminated by renal and
hepatic excretion. However, dose reduction is not required in geriatric
patients or patients with renal insufficiency. For patients with hepatic
insufficiency, dose reduction may be required with ondansetron.
5-HT3-receptor antagonists
do not inhibit dopamine or mus-carinic receptors. They do not have effects on
esophageal or gastric motility but may slow colonic transit.
Clinical Uses
A. Chemotherapy-Induced Nausea and Vomiting
5-HT3-receptor antagonists
are the primary agents for the preven-tion of acute chemotherapy-induced nausea
and emesis. When used alone, these drugs have little or no efficacy for the
prevention of delayed nausea and vomiting (ie, occurring > 24 hours after
chemotherapy). The drugs are most effective when given as a single dose by
intravenous injection 30 minutes prior to administration of chemotherapy in the
following doses: ondansetron, 8 mg; gran-isetron, 1 mg; dolasetron, 100 mg; or
palonosetron, 0.25 mg. A single oral dose given 1 hour before chemotherapy may
be equally effective in the following regimens: ondansetron 8 mg twice daily or
24 mg once; granisetron, 2 mg; dolasetron, 100 mg. Although 5-HT3-receptor antagonists
are effective as single agents for the prevention of chemotherapy-induced
nausea and vomiting, their efficacy is enhanced by combination therapy with a
corticosteroid (dexamethasone) and NK1-receptor antagonist .
B. Postoperative and Postradiation Nausea and Vomiting
5-HT3-receptor
antagonists are used to prevent or treat postoperative nausea and vomiting.
Because of adverse effects and increased restric-tions on the use of other
antiemetic agents, 5-HT3-receptor
antago-nists are increasingly used for this indication. They are also effective
in the prevention and treatment of nausea and vomiting in patients undergoing
radiation therapy to the whole body or abdomen.
Adverse Effects
The
5-HT3-receptor antagonists are
well-tolerated agents with excel-lent safety profiles. The most commonly
reported adverse effects are headache, dizziness, and constipation. All four
agents cause a small but statistically significant prolongation of the QT
interval, but this is most pronounced with dolasetron. Although cardiac
arrhythmias have not been linked to dolasetron, it should not be administered
to patients with prolonged QT or in conjunction with other medica-tions that
may prolong the QT interval .
Drug Interactions
No significant drug
interactions have been reported with 5-HT 3-receptor antagonists. All four agents undergo
some metabolism by the hepatic cytochrome P450 system but they do not appear
toaffect the metabolism of other drugs. However, other drugs may reduce hepatic
clearance of the 5-HT3-receptor antagonists, alter-ing their half-life.
CORTICOSTEROIDS
Corticosteroids
(dexamethasone, methylprednisolone) have antie-metic properties, but the basis
for these effects is unknown.. These agents appear to enhance the efficacy of
5-HT3-receptor antagonists for
prevention of acute and delayed nausea and vomiting in patients receiving
moderately to highly emetogenic chemotherapy regimens. Although a number of
corticosteroids have been used, dexame-thasone, 8–20 mg intravenously before
chemotherapy, followed by 8 mg/d orally for 2–4 days, is commonly administered.
NEUROKININ RECEPTOR ANTAGONISTS
Neurokinin 1 (NK1)-receptor antagonists
have antiemetic proper-ties that are mediated through central blockade in the
area pos-trema. Aprepitant (an oral
formulation) is a highly selective NK1-receptor antagonist that crosses the
blood-brain barrier and occupies brain NK1 receptors. It has no affinity for serotonin,
dopamine, or corticosteroid receptors. Fosaprepitant
is an intra-venous formulation that is converted within 30 minutes after
infusion to aprepitant.
Pharmacokinetics
The oral
bioavailability of aprepitant is 65%, and the serum half-life is 12 hours.
Aprepitant is metabolized by the liver, primarily by the CYP3A4 pathway.
Clinical Uses
Aprepitant is used in
combination with 5-HT3-receptor antago-nists and corticosteroids for the prevention of
acute and delayed nausea and vomiting from highly emetogenic chemotherapeutic
regimens. Combined therapy with aprepitant, a 5-HT3-receptor antagonist, and dexamethasone
prevents acute emesis in 80–90% of patients compared with less than 70% treated
without aprepi-tant. Prevention of delayed emesis occurs in more than 70% of
patients receiving combined therapy versus 30–50% treated with-out aprepitant.
NK1-receptor antagonists
may be administered for 3 days as follows: oral aprepitant 125 mg or
intravenous fosa-prepitant 115 mg given 1 hour before chemotherapy, followed by
oral aprepitant 80 mg/d for 2 days after chemotherapy.
Adverse Effects & Drug Interactions
Aprepitant may be
associated with fatigue, dizziness, and diarrhea. The drug is metabolized by
CYP3A4 and may inhibit the metabo-lism of other drugs metabolized by the CYP3A4
pathway. Several chemotherapeutic agents are metabolized by CYP3A4, including
docetaxel, paclitaxel, etoposide, irinotecan, imatinib, vinblastine, and
vincristine. Drugs that inhibit CYP3A4 metabolism maysignificantly increase
aprepitant plasma levels (eg, ketoconazole, ciprofloxacin, clarithromycin,
nefazodone, ritonavir, nelfinavir, verapamil, and quinidine). Aprepitant decreases
the international normalized ratio (INR) in patients taking warfarin.
PHENOTHIAZINES & BUTYROPHENONES
Phenothiazines are
antipsychotic agents that can be used for their potent antiemetic and sedative
properties . The antiemetic properties of phenothiazines are mediated through
inhibition of dopamine and muscarinic receptors. Sedative prop-erties are due
to their antihistamine activity. The agents most commonly used as antiemetics
are prochlorperazine, promethaz-ine, and thiethylperazine.
Antipsychotic butyrophenones
also possess antiemetic proper-ties due to their central dopaminergic blockade
. The main agent used is droperidol,
which can be given by intra-muscular or intravenous injection. In antiemetic
doses, droperidol is extremely sedating. Previously, it was used extensively
for post-operative nausea and vomiting, in conjunction with opiates and
benzodiazepines for sedation for surgical and endoscopic proce-dures, for
neuroleptanalgesia, and for induction and maintenance of general anesthesia.
Extrapyramidal effects and hypotension may occur. Droperidol may prolong the QT
interval, rarely resulting in fatal episodes of ventricular tachycardia
including torsades de pointes. Therefore, droperidol should not be used in
patients with QT prolongation and should be used only in patients who have not
responded adequately to alternative agents.
SUBSTITUTED BENZAMIDES
Substituted
benzamides include metoclopramide
(discussed pre-viously) and trimethobenzamide.
Their primary mechanism of antiemetic action is believed to be
dopamine-receptor blockade. Trimethobenzamide also has weak antihistaminic
activity. For prevention and treatment of nausea and vomiting, metoclo-pramide
may be given in the relatively high dosage of 10–20 mg orally or intravenously
every 6 hours. The usual dose of trimethobenzamide is 300 mg orally, or 200 mg
by intramuscular injection. The principal adverse effects of these central
dopamine antagonists are extrapyramidal: restlessness, dystonias, and
parkin-sonian symptoms.
H1
ANTIHISTAMINES & ANTICHOLINERGIC DRUGS
As single agents,
these drugs have weak antiemetic activity, although they are particularly
useful for the prevention or treatment of motion sickness. Their use may be
limited by dizziness, sedation, confusion, dry mouth, cycloplegia, and urinary
retention. Diphenhydramine and one
of its salts, dimenhydrinate, are
first-generation histamine H1 antagonists that also have significant
anticholinergic
properties. Because of its sedating properties, diphenhydramine is commonly
used in conjunction with other antiemetics for treatment of emesis due to
chemotherapy. Meclizine is an H1antihistaminic agent
with minimal anticholin-ergic properties that also causes less sedation. It is
used for the prevention of motion sickness and the treatment of vertigo due to
labyrinth dysfunction.
Hyoscine (scopolamine), a prototypic muscarinic receptorantagonist, is
one of the best agents for the prevention of motion sickness. However, it has a
very high incidence of anticholinergic effects when given orally or
parenterally. It is better tolerated as a transdermal patch. Superiority to
dimenhydrinate has not been proved.
BENZODIAZEPINES
Benzodiazepines such
as lorazepam or diazepam are used before the initiation of chemotherapy to
reduce anticipatory vomiting or vomiting caused by anxiety.
CANNABINOIDS
Dronabinol is9-tetrahydrocannabinol (THC), the major psy-choactive chemical in
marijuana . After oral ingestion, the drug is almost completely absorbed but
undergoes significant first-pass hepatic metabolism. Its metabolites are
excreted slowly over days to weeks in the feces and urine. Like crude
marijuana, dronabinol is a psychoactive agent that is used medically as an
appetite stimulant and as an antiemetic, but the mechanisms for these effects
are not understood. Because of the availability of more effective agents,
dronabinol now is uncom-monly used for the prevention of chemotherapy-induced
nausea and vomiting. Combination therapy with phenothiazines provides
synergistic antiemetic action and appears to attenuate the adverse effects of
both agents. Dronabinol is usually administered in a dosage of 5 mg/m2 just prior to
chemotherapy and every 2–4 hours as needed. Adverse effects include euphoria,
dysphoria, sedation, hallucinations, dry mouth, and increased appetite. It has
some autonomic effects that may result in tachycardia, conjunctival injection,
and orthostatic hypotension. Dronabinol has no signifi-cant drug-drug
interactions but may potentiate the clinical effects of other psychoactive
agents.
Nabilone is a closely related THC analog that has been avail-able in
other countries and is now approved for use in the USA.
Related Topics