Chapter: Basic & Clinical Pharmacology : Drugs Used in the Treatment of Gastrointestinal Diseases
Aminosalicylates
AMINOSALICYLATES
Chemistry & Formulations
Drugs that contain 5-aminosalicylic acid (5-ASA) have been
used successfully for decades in the treatment of inflammatory bowel diseases
(Figure 62–8). 5-ASA differs from salicylic acid only by the addition of an
amino group at the 5 (meta) position. Aminosalicylates are believed to work
topically (not systemically) in areas of diseased gastrointestinal mucosa. Up
to 80% of unfor-mulated, aqueous 5-ASA is absorbed from the small intestine and
does not reach the distal small bowel or colon in appreciable quan-tities. To
overcome the rapid absorption of 5-ASA from the proximal small intestine, a
number of formulations have been designed to deliver 5-ASA to various distal
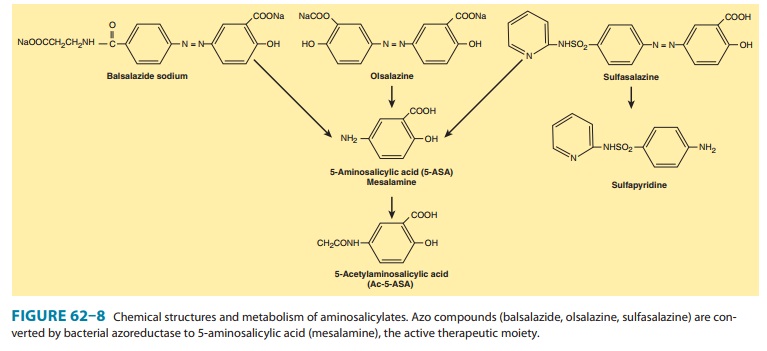
segments of the small bowel or the colon. These include sulfasalazine, olsalazine, bal-salazide, and various forms of mesalamine.
A. Azo Compounds
Sulfasalazine, balsalazide, and olsalazine contain 5-ASA bound by an azo (N=N) bond to an inert compound or to another 5-ASA molecule (Figure 62–8). In sulfasalazine, 5-ASA is bound to sul-fapyridine; in balsalazide, 5-ASA is bound to 4-aminobenzoyl-β-alanine; and in olsalazine, two 5-ASA molecules are bound together. The azo structure markedly reduces absorption of the parent drug from the small intestine. In the terminal ileum and colon, resident bacteria cleave the azo bond by means of an azore-ductase enzyme, releasing the active 5-ASA. Consequently, high concentrations of active drug are made available in the terminal ileum or colon.
B. Mesalamine Compounds
Other proprietary
formulations have been designed that package 5-ASA itself in various ways to
deliver it to different segments of the small or large bowel. These 5-ASA
formulations are known generically as mesalamine.
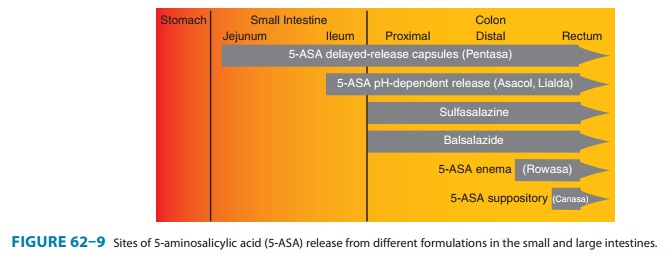
Pentasa is a mesalamine formulation that contains timed-release
microgranules that release 5-ASA throughout the small intestine (Figure 62–9). Asacol and Apriso have 5-ASA coated in a pH-sensitive resin that dissolves at
pH 6-7 (the pH of the distal ileum and proximal colon). Lialda also uses a pH-dependent resin that encases a multimatrix
core. On dissolu-tion of the pH-sensitive resin in the colon, water slowly
penetrates its hydrophilic and lipophilic core, leading to slow release of
mesalamine throughout the colon. 5-ASA also may be delivered in high
concentrations to the rectum and sigmoid colon by means of enema formulations (Rowasa) or suppositories (Canasa).
Pharmacokinetics & Pharmacodynamics
Although unformulated
5-ASA is readily absorbed from the small intestine, absorption of 5-ASA from
the colon is extremely low. In contrast, approximately 20–30% of 5-ASA from
current oral mesalamine formulations is systemically absorbed in the small intestine.
Absorbed 5-ASA undergoes N-acetylation
in the gut epithelium and liver to a metabolite that does not possess
signifi-cant anti-inflammatory activity. The acetylated metabolite is excreted
by the kidneys.
Of the azo compounds,
10% of sulfasalazine and less than 1% of balsalazide are absorbed as native
compounds. After azore-ductase breakdown of sulfasalazine, over 85% of the
carrier mol-ecule sulfapyridine is absorbed from the colon. Sulfapyridine
undergoes hepatic metabolism (including acetylation) followed by renal
excretion. By contrast, after azoreductase breakdown of bal-salazide, over 70%
of the carrier peptide is recovered intact in the feces and only a small amount
of systemic absorption occurs.
The mechanism of
action of 5-ASA is not certain. The primary action of salicylate and other
NSAIDs is due to blockade ofprostaglandin synthesis by inhibition of
cyclooxygenase. However, the aminosalicylates have variable effects on
prostaglandin produc-tion. It is thought that 5-ASA modulates inflammatory
mediators derived from both the cyclooxygenase and lipoxygenase pathways. Other
potential mechanisms of action of the 5-ASA drugs relate to their ability to
interfere with the production of inflammatory cytokines. 5-ASA inhibits the
activity of nuclear factor-κB (NF-κB), an important
transcription factor for proinflammatory cytokines. 5-ASA may also inhibit
cellular functions of natural killer cells, mucosal lymphocytes, and
macrophages, and it may scavenge reactive oxygen metabolites.
Clinical Uses
5-ASA drugs induce and
maintain remission in ulcerative colitis and are considered to be the
first-line agents for treatment of mild to moderate active ulcerative colitis.
Their efficacy in Crohn’s disease is unproven, although many clinicians use
5-ASA agents as first-line therapy for mild to moderate disease involving the
colon or distal ileum.
The effectiveness of
5-ASA therapy depends in part on achiev-ing high drug concentration at the site
of active disease. Thus, 5-ASA suppositories or enemas are useful in patients
with ulcer-ative colitis or Crohn’s disease confined to the rectum (proctitis)
or distal colon (proctosigmoiditis). In patients with ulcerative colitis or
Crohn’s colitis that extends to the proximal colon, both the azo compounds and
mesalamine formulations are useful. For the treatment of Crohn’s disease
involving the small bowel, mesal-amine compounds, which release 5-ASA in the
small intestine, have a theoretic advantage over the azo compounds.
Adverse Effects
Sulfasalazine has a
high incidence of adverse effects, most of which are attributable to systemic
effects of the sulfapyridine molecule. Slow acetylators of sulfapyridine have
more frequent and more severe adverse effects than fast acetylators. Up to 40%
of patients cannot tolerate therapeutic doses of sulfasalazine. The most
com-mon problems are dose-related and include nausea, gastrointestinal
upset, headaches, arthralgias, myalgias, bone marrow suppression, and malaise.
Hypersensitivity to sulfapyridine (or, rarely, 5-ASA) can result in fever,
exfoliative dermatitis, pancreatitis, pneumoni-tis, hemolytic anemia,
pericarditis, or hepatitis. Sulfasalazine has also been associated with
oligospermia, which reverses upon dis-continuation of the drug. Sulfasalazine
impairs folate absorption and processing; hence, dietary supplementation with 1
mg/d folic acid is recommended.
In contrast to
sulfasalazine, other aminosalicylate formulations are well tolerated. In most
clinical trials, the frequency of drug adverse events is similar to that in patients
treated with placebo. For unclear reasons, olsalazine may stimulate a secretory
diar-rhea—which should not be confused with active inflammatory bowel
disease—in 10% of patients. Rare hypersensitivity reactions may occur with all
aminosalicylates but are much less common than with sulfasalazine. Careful
studies have documented subtle changes indicative of renal tubular damage in
patients receiving high doses of aminosalicylates. Rare cases of interstitial
nephritis are reported, particularly in association with high doses of
mesala-mine formulations; this may be attributable to the higher serum 5-ASA
levels attained with these drugs. Sulfasalazine and other aminosalicylates
rarely cause worsening of colitis, which may be misinterpreted as refractory
colitis.
Related Topics