Chapter: Plant Biochemistry: The use of energy from sunlight by photosynthesis is the basis of life on earth
An antenna is required to capture light
An antenna is required to capture light
In order to excite a photosynthetic reaction center, a photon with defined energy content has to react with a chlorophyll molecule in the reaction center. The probability is very low that a photon not only has the proper energy, but also hits the pigment exactly at the site of the chlorophyll mol-ecule. Therefore efficient photosynthesis is possible only when the energy of photons of various wavelengths is captured over a certain surface by a so-called antenna (Fig. 2.8). Similarly, radio and television sets could not work without an antenna.
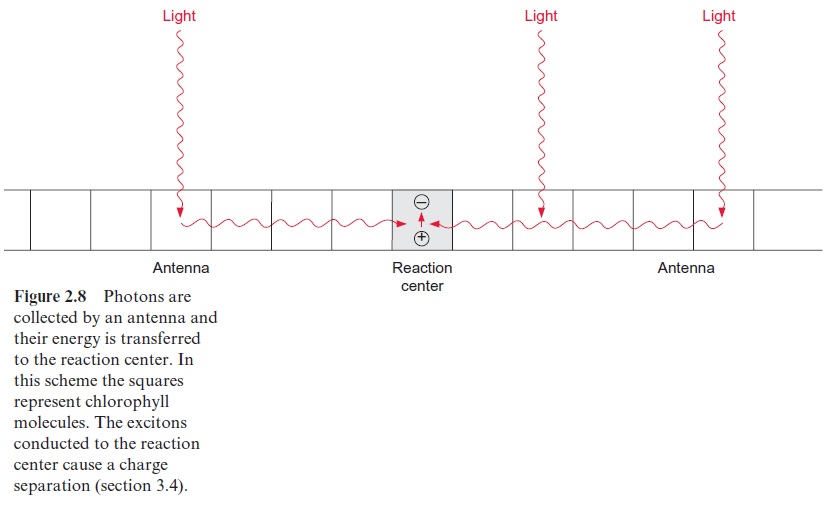
The antennae of plants consist of a large number of protein-bound chlorophyll molecules that absorb photons and transfer their energy to the reaction center. Only a few thousandths of the chlorophyll molecules in the leaf are constituents of the actual reaction centers; the remainder are contained in the antennae. Observations made as early as 1932 by Robert Emerson and William Arnold in the United States indicated that the large majority of chlorophyll molecules are not part of the reaction centers. The two researchers illuminated a suspension of the green alga Chlorella with light pulses of 10 s duration, interrupted by dark intervals of 20 ms. Evolution of oxygen was used as a measure for photosynthesis. The light pulses were made so short that chlorophyll could undergo only one photo-synthetic excitation cycle and a high light intensity was chosen in order to achieve maximum oxygen evolution. Apparently the photosynthetic appa-ratus was thus saturated with photons. Analysis of the chlorophyll content of the algae suspension showed that under saturating conditions only one molecule of O2 was formed per 2,400 chlorophyll molecules.
In the following years Robert Emerson refined these experiments and was able to show when pulses were applied at very low light intensity, the amount of oxygen formed increased proportionally with the light intensity. From this it was calculated that the release of one molecule of oxygen had a minimum quantum requirement of about eight photons. These results set-tled a long scientific dispute with Otto Warburg, who had concluded from his experiments that only four photons are required for the evolution of one molecule of O2. Later it was recognized that each of the two reaction cent-ers requires four photons for the formation of O2. Moreover, the results of Emerson and Arnold allowed the calculation that about 300 chlorophyll molecules are associated with one reaction center. These are constituents of the antennae.
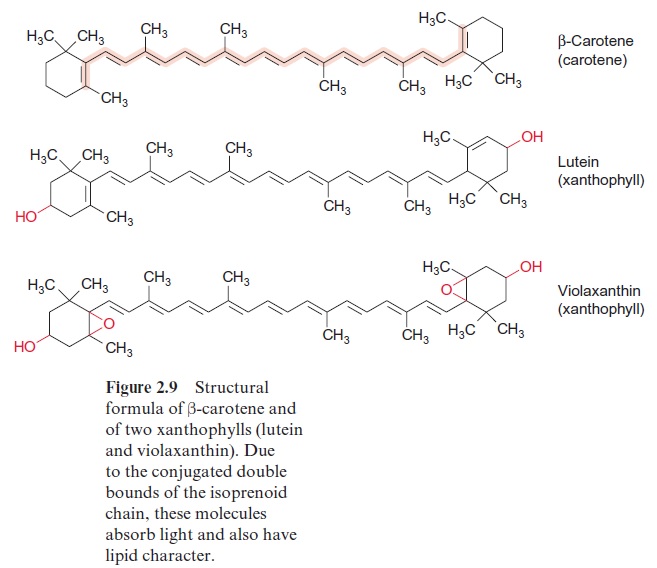
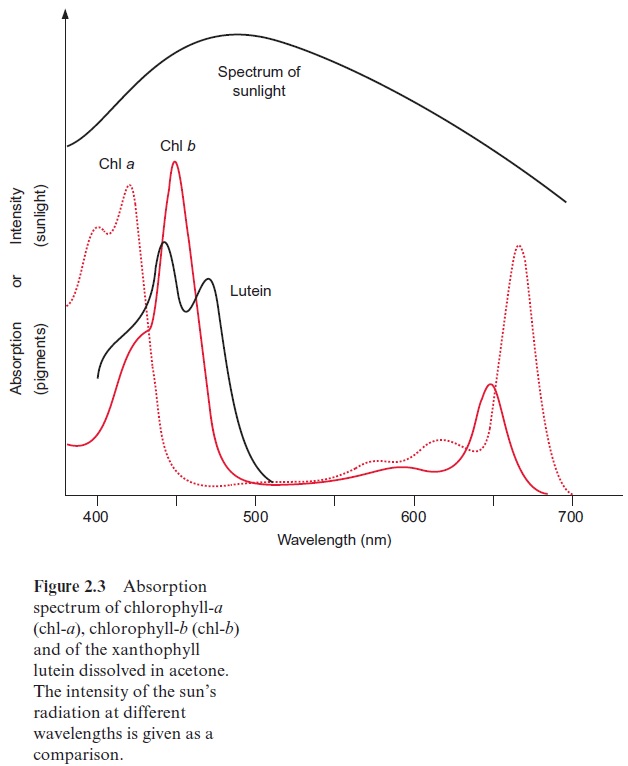
The antennae contain additional accessory pigments to utilize those photons where the wavelength corresponds to the “green window” between the absorption maxima of the chlorophylls. In higher plants these pigments are carotenoids, mainly xanthophylls, including lutein and the related vio-laxanthin as well as carotenes such as -carotene to name the major com-pound (Fig. 2.9). Moreover, an important function of these carotenoids in the antennae is to prevent the formation of the harmful triplet state of the chlorophylls. Important constituents of the antennae in cyanobacteria are phycobilins.
How is the excitation energy of the photons captured in the antennae and transferred to the reaction centers?
The transfer of energy in the antennae via electron transport from chromophore to chromophore in a sequence of redox processes, as in the elec-tron transport chains of photosynthesis or of mitochondrial respiration, could be excluded, since such an electron transport would need considerable activation energy. This is not the case, since a flux of excitation energy can be measured in the antennae at temperatures as low as 1 K. At these low temperatures light absorption and fluorescence still occur, whereas chemical processes catalyzed by enzymes are completely inactive. Under these conditions the energy transfer in the antennae pro-ceeds according to a mechanism that is related to those of light absorption and fluorescence.
When chromophores are positioned very close to each other, the quan-tum energy of an irradiated photon is transferred from one chromophore to the next. One quantum of light energy is named a photon, one quantum of excitation energy transferred from one molecule to the next is termed an exciton. A prerequisite for the transfer of excitons is a specific position-ing of the chromophores. This is arranged by proteins, and therefore the chromophores of the antennae always occur as protein complexes.
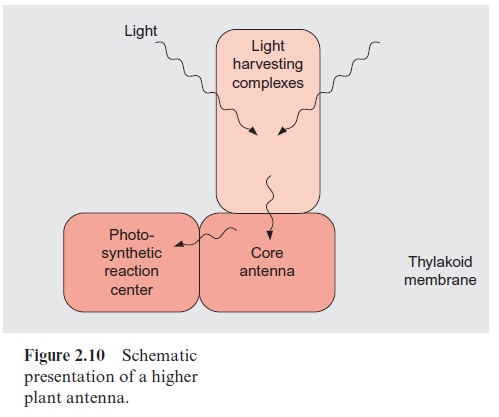
The antennae of plants consist of an inner part and an outer part (Fig. 2.10). The outer antenna, formed by the light harvesting complexes (LHCs), col-lects the light. The inner antenna, consisting of the core complexes, is an integral constituent of the reaction centers; it also collects light and con-ducts the excitons that were collected in the outer antenna to the photosyn-thetic reaction centers.
The LHCs are composed of polypeptides, which bind chl-a, chl-b, xan-thophylls, and carotenes. These proteins, termed LHC polypeptides, are encoded in the nucleus. A plant contains many different LHC polypeptides. In a tomato, for instance, at least 19 different genes for LHC polypeptides have been found, which are very similar to each other and are members of a multigene family. They are homologous, as they have all evolved from a common ancestor.
Plants contain two reaction centers, which are arranged in sequence: a reaction center of photosystem II (PS II), which has an absorption maxi-mum at 680 nm, and a photosystem I (PS I) with an absorption maximum at 700 nm. The function of these reaction centers will be described in sec-tions 3.6 and 3.8. Both photosystems are composed of different LHCs.
The function of an antenna is illustrated by the antenna of photosystem II
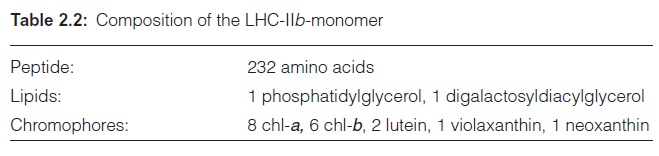
The antenna of the PS II reaction center contains primarily four LHCs termed LHC-IIa–d. The main component is LHC-IIb; it represents 67% of the total chlorophyll of the PS II antenna and is the most abundant mem-brane protein of the thylakoid membrane, and has therefore been partic-ularly thoroughly investigated. LHC-IIb occurs in the membrane, most probably as a trimer. The monomer consists of a polypeptide to which four xanthophyll molecules are bound (Table 2.2). The polypeptide contains one threonine residue, which can be phosphorylated by ATP via a protein kinase. Phosphorylation regulates the activity of LHC-II .
There has been a breakthrough in establishing the three-dimensional structure of LHC-IIb by electron cryomicroscopy at a temperature of 4 K of crystalline layers of LHC-IIb-trimers (Fig. 2.11). The LHC-IIb-peptide forms three transmembrane helices. The two lutein molecules span the membrane crosswise. The other two molecules are not visible in the iso-lated LHC complex. The chl-b-molecules, where the absorption maximum in the red spectral region lies at a shorter wavelength than that of chl-a, are positioned in the outer region of the complexes. Only one of the chl-a-molecules is positioned in the outer region; the others are all present in the center. Figure 2.12 shows a vertical projection of the arrangement of the monomers to form a trimer. The chl-a positioned in the outer region medi-ates the transfer of energy to the neighboring trimers or to the reaction center. The trimers are arranged in the membrane as oligomers forming the antenna for the conductance of the absorbed excitons. The chl-a/chl-bratio is much higher in LHC-IIa and LHC-IIc than in LHC-IIb. Most likely LHC-IIa and LHC-IIc are positioned between LHC-IIb and the reaction center.
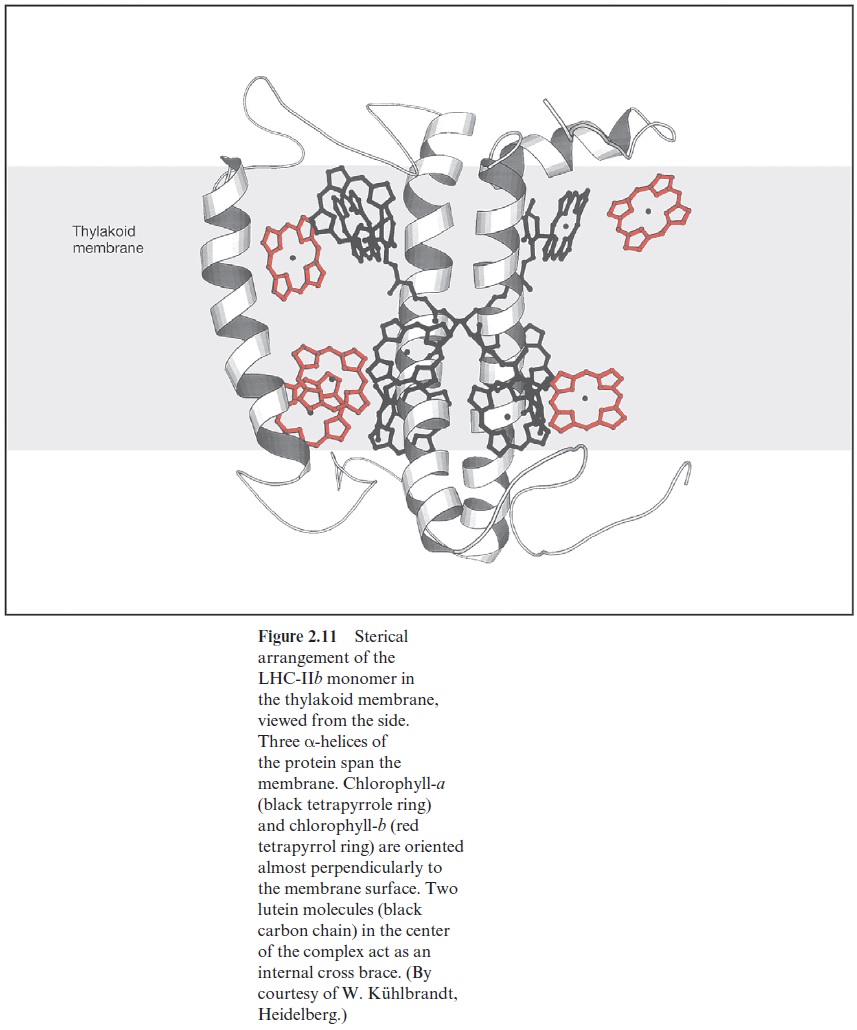

Figure 2.13 shows a hypothetical scheme of the array of the PS II antenna. The outer complexes, consisting of LHC-IIb, are present at the periphery of the antenna. The excitons captured by chl-b in LHC-IIb are transferred to chl-a in the center of the LHC-IIb monomers and are then transferred further by chl-a contacts between the trimers to the inner anten-nae complexes. The inner complexes are connected by small chlorophyll containing subunits to the core complex. This consists of the antennae pro-teins CP 43 and CP 47, which are closely attached to the reaction center (Fig. 3.22), and each containing about 15 chl-a molecules. Since the absorp-tion maximum of chl-b is at a lower wavelength than that of chl-a, the transfer of excitons from chl-b to chl-a is accompanied by loss of energy as heat. This promotes the flux of excitons from the periphery to the reaction center. The connection between the outer LHCs (LHC-IIb) and the PS II can be interrupted by phosphorylation. In this way the actual size of the antenna can be adjusted to the intensity of illumination .
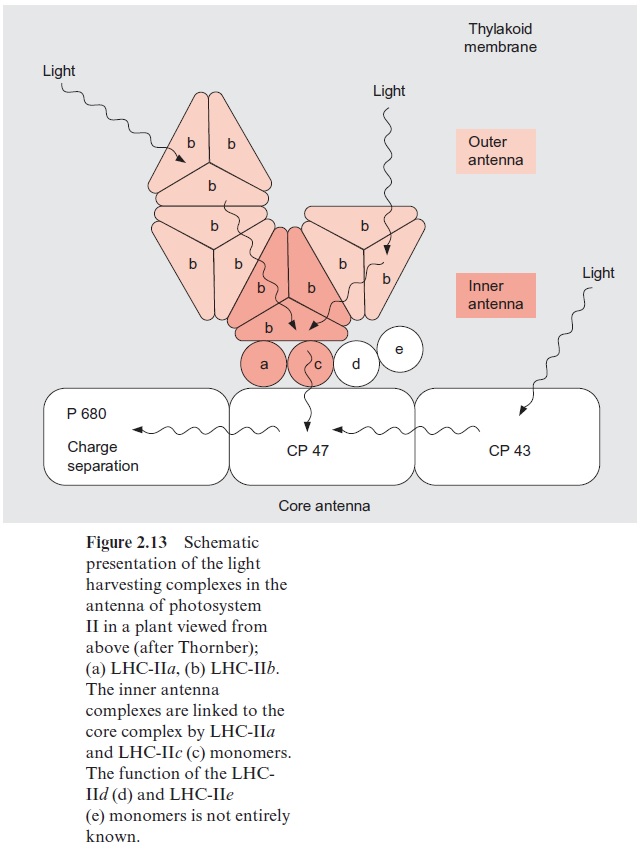
Photosystem I contains fewer LHCs than photosystem II since its core antenna is larger than in PS II. The LHCs of PS I are similar to those of PS II. Sequence analysis shows that LHC-I and LHC-II stem from a common ancestor. It has been suggested that in the phosphorylated state LHC-IIb can also function as an antenna of PS I .
There are two mechanisms for the movement of excitons. The excitons may be delocalized by distribution over the whole chromophore mole-cules. On the other hand, excitons may also initially be present in a certain chromophore molecule and subsequently transferred to a more distant chromophore. This process of exciton transfer has been termed the Förster mechanism. The transfer of excitons between closely neighboring chlorophyll molecules within an LHC complex probably proceeds viadelocalized electrons, and the transfer between the LHCs and the reaction center occurs via the Förster mechanism. Absorption measurements with ultrafast laser technique have shown that the exciton transfer between two chlorophyll molecules proceeds within 0.1 ps (10-13 s). Thus the velocity of the exciton transfer in the antennae is much faster than the charge separation in the reaction center (approx 3.5 ps) . The reaction center functions as an energy trap for excitons present in the antenna.
Phycobilisomes enable cyanobacteria and red algae to carry out photosynthesis even in dim light
Cyanobacteria and red algae possess antennae structures that can collect light of very low intensity. These antennae are arranged as complexes on top of the membrane near the reaction centers of photosystem II (Fig. 2.14).
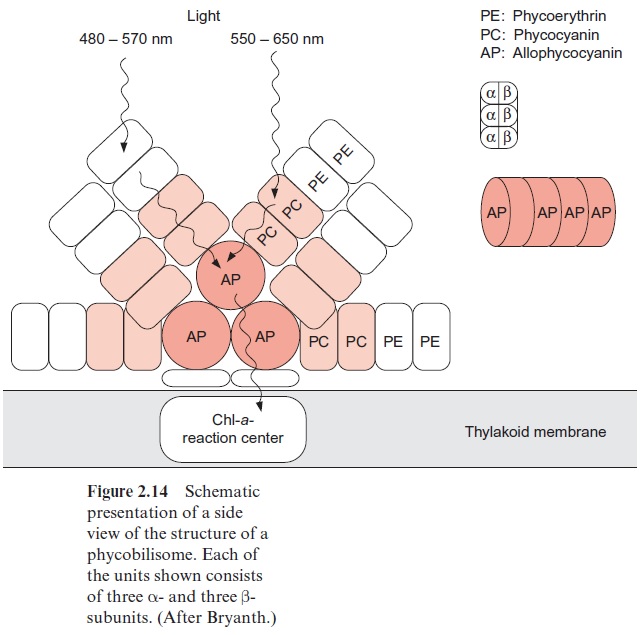
These complexes, termed phycobilisomes, consist of proteins (phyco-biliproteins), which are covalently linked with phycobilins. Phycobilins are open-chained tetrapyrroles and therefore are structurally related to the chlorophylls. Open-chained tetrapyrroles are also contained in bile, which explains the name -bilin. The phycobilins are linked to the protein by a thioether bond between an SH-group of the protein and the vinyl side chain of the phycobilin. The protein phycoerythrin is linked to the chromophore phycoerythrobilin, and the proteins phycocyanin and allophycocyanin to the chromophore phycocyanobilin (Fig. 2.15). The basic structure in the phyco-biliproteins consists of a heterodimer composed of α- and β-subunits. Each of these protein subunits binds one to four phycobilins as a chromophore. Three of these heterodimers aggregate to a trimer ( α‚β )3 and thus form the actual building block of a phycobilisome. Specific linker polypeptides func-tion as “mortar” between the building blocks.
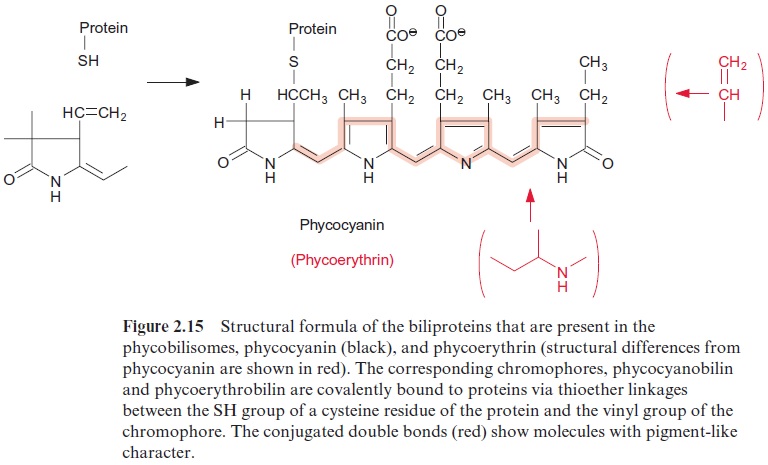
Figure 2.14 shows the structure of a phycobilisome. The phycobilisome is attached to the membrane by anchor proteins. Three aggregates of four to five ( α,β )3 units form the core. This core contains the chromophore allo-phycocyanin (AP) to which cylindrical rod like structures are attached, each with four to six building blocks. The inner units contain mainly phycocyanine (PC) and the outer ones phycoerythrin (PE). The function of this structural organization is illustrated by the absorption spectra of the various biliproteins shown in Figure 2.16. The light of shorter wave-length is absorbed in the periphery of the rods by phycoerythrin and the light of longer wavelength in the inner regions of the rods by phycocyanin. The core transfers the excitons to the reaction center. The principle of spa-tial distribution between the short wavelength absorbing pigments at the periphery and the long wavelength absorbing pigments in the center is also implemented for the PS II antennae of higher plants (Fig. 2.10).
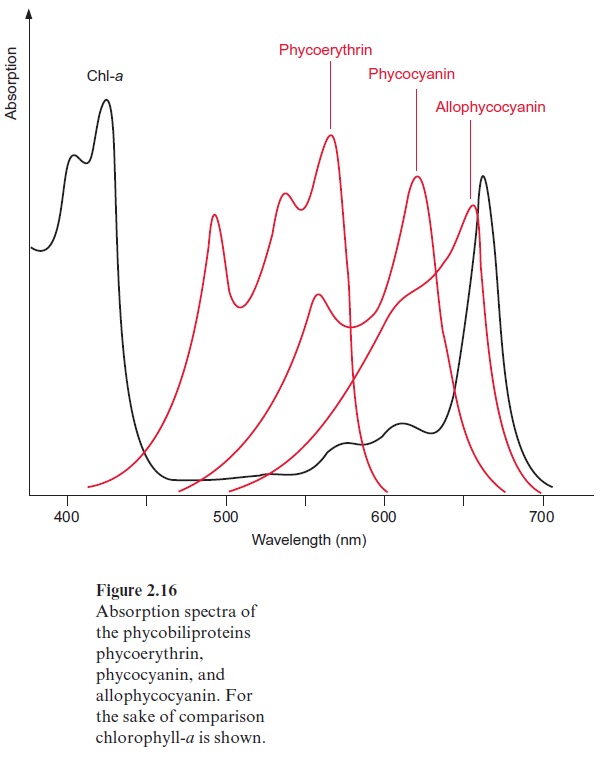
Due to the phycobiliproteins, phycobilisomes are able to absorb green light very efficiently (Fig. 2.16), thus allowing cyanobacteria and red algae to survive in deep waters with low light intensities. At these depths, due to the “green window” of photosynthesis (Fig. 2.3), only green light is avail-able, as the light of the other wavelengths is absorbed by green algae living in the upper regions of the water column. The algae in the deeper regions are obliged to invest a large portion of their cellular matter in phycobili-somes in order to carry out photosynthesis at this very low light intensity and at distinct wavelengths. Biliproteins can amount to 40% of the total cellular protein of the algae. These organisms undertake an extraordinary expenditure to collect enough light for survival.
Related Topics