Chapter: Basic & Clinical Pharmacology : Nonsteroidal Anti-Inflammatory Drugs, Disease-Modifying Antirheumatic Drugs, Nonopioid Analgesics,& Drugs Used in Gout
Allopurinol
ALLOPURINOL
The
preferred and standard-of-care therapy for gout during the period between acute
episodes is allopurinol, which reduces total uric acid body burden by
inhibiting xanthine oxidase.
Chemistry
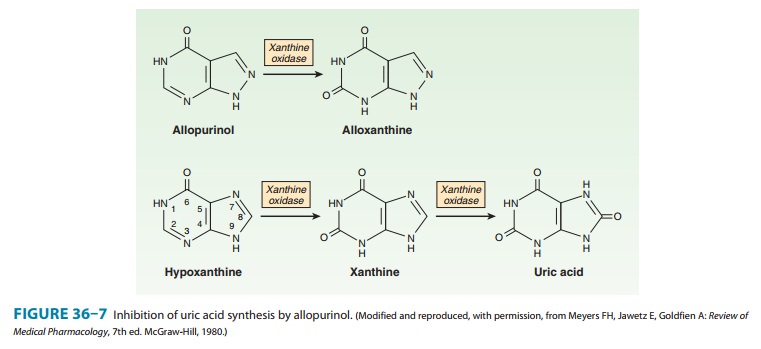
The structure of
allopurinol, an isomer of hypoxanthine, is shown in Figure 36–7.
Pharmacokinetics
Allopurinol
is approximately 80% absorbed after oral administra-tion and has a terminal
serum half-life of 1–2 hours. Like uric acid, allopurinol is metabolized by
xanthine oxidase, but the resulting compound, alloxanthine, retains the
capacity to inhibit xanthine oxidase and has a long enough duration of action
so that allopurinol is given only once a day.
Pharmacodynamics
Dietary
purines are not an important source of uric acid. Quantitatively important
amounts of purine are formed from amino acids, formate, and carbon dioxide in
the body. Those purine ribonucleotides not incorporated into nucleic acids and
derived from nucleic acid degradation are converted to xanthine or hypoxanthine
and oxidized to uric acid (Figure 36–7). Allopurinol inhibits this last step,
resulting in a fall in the plasma urate level and a decrease in the overall urate
burden. The more soluble xanthine and hypoxanthine are increased.
Indications
Allopurinol is often the first-line agent for the treatment of chronic gout in the period between attacks and it aims to prolong the intercritical period.
As with uricosuric agents, the therapy is begun with the
expectation that it will be continued for years if not for life. When
initiating allopurinol, colchicine or NSAID should be used until steady-state
serum uric acid is normalized or decreased to less than 6 mg/dL and they should
be continued for 3-6 months or even longer if required. Thereafter, colchicine
or the NSAID can be cautiously stopped while continuing allopurinol therapy. In
addition to gout, allopurinol is also used as an antipro-tozoal agent and is indicated to prevent the mas-sive
uricosuria following therapy of blood dyscrasias that could otherwise lead to
renal calculi.
Adverse Effects
See above for
prophylaxis against an acute attack during the initia-tion of allopurinol,
which can occur as a result of acute changes in the serum uric acid level.
Among the side effects, GI intolerance including nausea, vomiting, and
diarrhea, peripheral neuritis and necrotizing vasculitis, bone marrow
suppression, and rarely aplastic anemia may also occur. Hepatic toxicity and
interstitial nephritis have been reported. An allergic skin reaction
characterized by pru-ritic maculopapular lesions occurs in 3% of patients.
Isolated cases of exfoliative dermatitis have been reported. In very rare
cases, allopurinol has become bound to the lens, resulting in cataracts.
Interactions & Cautions
When
chemotherapeutic purines (eg, azathioprine) are given con-comitantly with
allopurinol, their dosage must be reduced by about 75%. Allopurinol may also
increase the effect of cyclophosphamide. Allopurinol inhibits the metabolism of
probenecid and oral antico-agulants and may increase hepatic iron
concentration. Safety in children and during pregnancy has not been
established.
Dosage
The initial dosage of
allopurinol is 100 mg/d. It should be titrated upward until serum uric acid is
below 6 mg/dL; this level is com-monly achieved at 300 mg/d but is not
restricted to this dose; doses as high as 800 mg/d may be needed.
As noted above,
colchicine or an NSAID should be given dur-ing the first several weeks of
allopurinol therapy to prevent the gouty arthritis episodes that sometimes
occur.
Related Topics