Chapter: Medicine and surgery: Haematology and clinical Immunology
Acute myeloid leukaemia - Acute leukaemia
Acute myeloid leukaemia
Definition
Malignant expansion of cells from the myeloid cell line.
Age
Most common in the middle aged and elderly
Sex
M = F
Aetiology
Aetiology is largely unknown although previous exposure to radiation, benzene or chemotherapy are known to be precipitating factors.
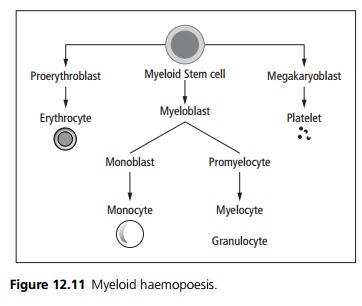
FAB sub classification (see Fig. 12.11) M0 Undifferentiated myeloblasts
M1 Myelocytic leukaemia without differentiation
M2 Myelocytic leukaemia with differentiation
M3 Acute promyelocytic leukaemia
M4 Acute myelomonocytic leukaemia
M5 Acute monocytic leukaemia proliferation of monoblasts
M6 Acute erythroblastic leukaemia (myeloblasts & proerythroblasts)
M7 Acute megakaryocytic leukaemia – myeloblasts & megakaryoblasts (uncommon)
Patients with M3 (acute promyelocytic leukaemia) are particularly prone to disseminated intravascular coagulation due to the presence of procoagulants within the cytoplasmic granules of the promyelocyte. In M3 there is a characteristic chromosomal translocation t(15:17), the promyelocytic leukaemia (PML) gene and the retinoic acid receptor come to lie next to each other and are therefore deregulated. Ninety-five per cent of patients with M3 are induced into remission by treatment with high dose retinoic acid. This is not sustained and chemotherapy should also be used.
Cells in M5 (acute monocytic leukaemia) secrete lysozyme (an antibacterial enzyme) which can damage the renal tubules causing hypokalaemia. Gum hypertrophy and hepatosplenomegaly is common within this subgroup.
Clinical features
Often there is an insidious onset of anorexia, malaise and lethargy due to anaemia. There is often a history of recurrent infections and/or easy bruising and mucosal bleeding. Other presentations include lymph node en-largement, bone and joint pain. On examination there may be pallor, bruising, hepatosplenomegaly and lymphadenopathy.
Microscopy
Abnormal leukaemic cells of the myeloid cell line replace the normal marrow. Morphologically the blast cells in AML may contain a stick like inclusion – Auer rods. The leukaemia is typed by cytochemical staining and monoclonal antibodies to look for cell surface markers.
Investigations
Blasts are seen in a peripheral blood film. Full blood count shows a low haemoglobin, variable white count, low platelet count. Bone marrow aspiration shows increased cellularity with a high percentage of the abnormal cells. Bone marrow cytogentic studies allow classification into prognostic groups (e.g. t(8:21) and inv 16 are good prognostic indicators, whereas −7 and −5 and poor prognostic indicators).
Management
High dose cytotoxic drugs doxorubicine, cytosine, etoposide are given cyclically. Supportive treatments include red blood cell transfusions, platelet transfusions and broad-spectrum antibiotics. Bone marrow transplantation may be used.
Prognosis
70% of those under 60 years will achieve remission with combination chemotherapy although the majority relapse within 3 years.
Related Topics