Chapter: Medical Physiology: Regulation of Acid-Base Balance
Use of Anion Gap to Diagnose Acid-Base Disorders
Use of Anion Gap to Diagnose Acid-Base Disorders
The concentrations of anions and cations in plasma must be equal to maintain electrical neutrality. There-fore, there is no real “anion gap” in the plasma. However, only certain cations and anions are routinely measured in the clinical laboratory. The cation normally measured is Na+, and the anions are usually Cl– and HCO3–. The “anion gap” (which is only a diagnostic concept) is the difference between unmeasured anions and unmeasured cations, and is estimated as
Plasma anion gap = [Na+] – [HCO3–] – [Cl–]
= 144 – 24 – 108 = 10 mEq/L
The anion gap will increase if unmeasured anions rise or if unmeasured cations fall. The most important unmeasured cations include calcium, magnesium, and potassium, and the major unmeasured anions are albumin, phosphate, sulfate, and other organic anions. Usually the unmeasured anions exceed the unmeasured cations, and the anion gap ranges between 8 and 16 mEq/L.
The plasma anion gap is used mainly in diagnosing different causes of metabolic acidosis. In metabolic acidosis, the plasma HCO3– is reduced. If the plasma sodium concentration is unchanged, the concentration of anions (either Cl– or an unmeasured anion) must increase to maintain electroneutrality. If plasma Cl– increases in proportion to the fall in plasma HCO3–, the anion gap will remain normal, and this is often referred to as hyperchloremic metabolic acidosis.
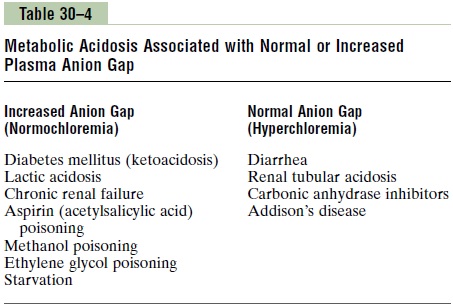
If the decrease in plasma HCO3– is not accompanied by increased Cl–, there must be increased levels of unmeasured anions and therefore an increase in the cal-culated anion gap. Metabolic acidosis caused by excess nonvolatile acids (besides HCl), such as lactic acid or ketoacids, is associated with an increased plasma anion gap because the fall in HCO3– is not matched by an equal increase in Cl–. Some examples of metabolic aci-dosis associated with a normal or increased anion gap are shown in Table 30–4. By calculating the anion gap, one can narrow some of the potential causes of meta-bolic acidosis.
Related Topics