Chapter: Basic & Clinical Pharmacology : Heavy Metal Intoxication & Chelators
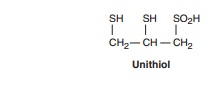
Unithiol (Dimercaptopropanesulfonic Acid, DMPS)
UNITHIOL (DIMERCAPTOPROPANESULFONIC
ACID, DMPS)
Unithiol, a dimercapto
chelating agent that is a water-soluble analog of dimercaprol, has been
available in the official formularies of Russia and other former Soviet
countries since 1958 and in Germany since 1976. It has been legally available
from com-pounding pharmacies in the USA since 1999. Unithiol can be
administered orally and intravenously. Bioavailability by the oral route is
approximately 50%, with peak blood levels occurring in approximately 3.7 hours.
Over 80% of an intravenous dose is excreted in the urine, mainly as cyclic DMPS
sulfides. The elimi-nation half-time of total unithiol (parent drug and its
transforma-tion products) is approximately 20 hours. Unithiol exhibits
protective effects against the toxic action of mercury and arsenic in animal
models, and it increases the excretion of mercury, arse-nic, and lead in
humans. Animal studies and a few case reports suggest that unithiol may also
have usefulness in the treatment of poisoning by bismuth compounds.
Indications & Toxicity
Unithiol
has no FDA-approved indications, but experimental studies and its pharmacologic
and pharmacodynamic profile suggest that intravenous unithiol offers advantages
over intra-muscular dimercaprol or oral succimer in the initial treatment of
severe acute poisoning by inorganic mercury or arsenic. Aqueous preparations of
unithiol (usually 50 mg/mL in sterile water) can be administered at a dosage of
3–5 mg/kg every 4 hours by slow intravenous infusion over 20 minutes. If a few
days of treatment are accompanied by stabilization of the patient’s
cardiovascular and gastrointestinal status, it may be possible to change to
oral administration of 4–8 mg/kg every 6–8 hours. Oral unithiol may also be
considered as an alterna-tive to oral succimer in the treatment of lead
intoxication.
Unithiol has been
reported to have a low overall incidence of adverse effects (< 4%). Self-limited
dermatologic reactions (drug exanthems or urticaria) are the most commonly
reported adverse effects, although isolated cases of major allergic reactions,
includ-ing erythema multiforme and Stevens-Johnson syndrome, have been
reported. Because rapid intravenous infusion may cause vasodilation and
hypotension, unithiol should be infused slowly over an interval of 15–20
minutes.
Related Topics