Chapter: Basic & Clinical Pharmacology : Heavy Metal Intoxication & Chelators
Penicillamine (D-Dimethlcysteine)
PENICILLAMINE (D-DIMETHLCYSTEINE)
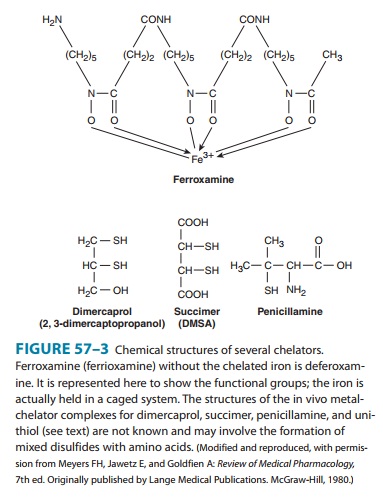
Penicillamine (Figure
57–3) is a white crystalline, water-soluble derivative of penicillin. D-Penicillamine is less toxic than the L
isomer and consequently is the preferred therapeutic form. Penicillamine is
readily absorbed from the gut and is resistant to metabolic degradation.
Indications & Toxicity
Penicillamine
is used chiefly for treatment of poisoning with cop-per or to prevent copper
accumulation, as in Wilson’s disease (hepatolenticular degeneration). It is
also used occasionally in the treatment of severe rheumatoid arthritis . Its
abil-ity to increase urinary excretion of lead and mercury had occa-sioned its
use in outpatient treatment for intoxication with these metals, but succimer,
with its stronger metal-mobilizing capacity and lower adverse-effect profile,
has generally replaced pen-icillamine for these purposes.
Adverse
effects have been seen in up to one third of patients receiving penicillamine.
Hypersensitivity reactions include rash, pruritus, and drug fever, and the drug
should be used with extreme caution, if at all, in patients with a history of
penicillin allergy. Nephrotoxicity with proteinuria has also been reported, and
protracted use of the drug may result in renal insufficiency. Pancytopenia has
been associated with prolonged drug intake. Pyridoxine deficiency is a frequent
toxic effect of other forms of the drug but is rarely seen with the D
form. An acetylated deriva-tive, N-acetylpenicillamine,
has been used experimentally in mer-cury poisoning and may have superior
metal-mobilizing capacity, but it is not commercially available.
Related Topics