Chapter: Basic & Clinical Pharmacology : Heavy Metal Intoxication & Chelators
Deferoxamine
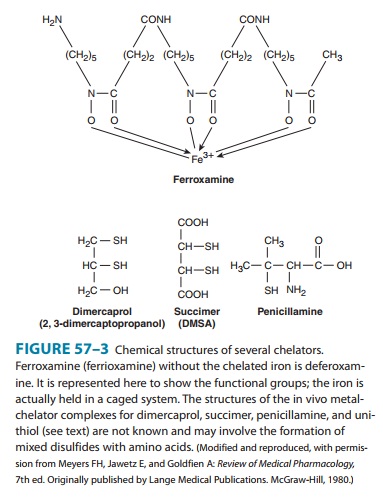
DEFEROXAMINE
Deferoxamine
is isolated from Streptomyces pilosus.
It binds iron avidly (Figure 57–3) but binds essential trace metals poorly.
Furthermore, though competing for loosely bound iron in iron-carrying proteins
(hemosiderin and ferritin), it fails to compete for biologically chelated iron,
as in microsomal and mitochondrial cytochromes and hemoproteins. Consequently,
it is the parenteral chelator of choice for iron poisoning. Deferoxamine plus
hemodialysis may also be useful in the treat-ment of aluminum toxicity in renal
failure. Deferoxamine is poorly absorbed when administered orally and may
increase iron absorption when given by this route. It should therefore be
admin-istered intramuscularly or, preferably, intravenously. It is believed to
be metabolized, but the pathways are unknown. The iron-chelator complex is
excreted in the urine, often turning the urine an orange-red color.
Rapid
intravenous administration may result in hypotension. Adverse idiosyncratic
responses such as flushing, abdominal discomfort, and rash have also been
observed. Pulmonary compli-cations (eg, acute respiratory distress syndrome)
have been reported in some patients undergoing deferoxamine infusions last-ing
longer than 24 hours, and neurotoxicity and increased suscep-tibility to
certain infections (eg, with Yersinia
enterocolitica) have been described after long-term therapy of iron
overload conditions (eg, thalassemia major).
Related Topics