Chapter: Environmental Biotechnology: Microbes and Metabolism
The dark reactions
The dark reactions
The result of illumination of a photosynthetic organism is to stimulate electron transport which leads to the production of NADPH or NADH, and synthesis of ATP. Both are required for the next stage which in eukaryotes and cyanobacteria (blue-green algae) is the synthesis of sugar from carbon dioxide involving the Calvin cycle. Many biochemistry textbooks give excellent descriptions of this process and so only a summary is given in Figure 2.10.
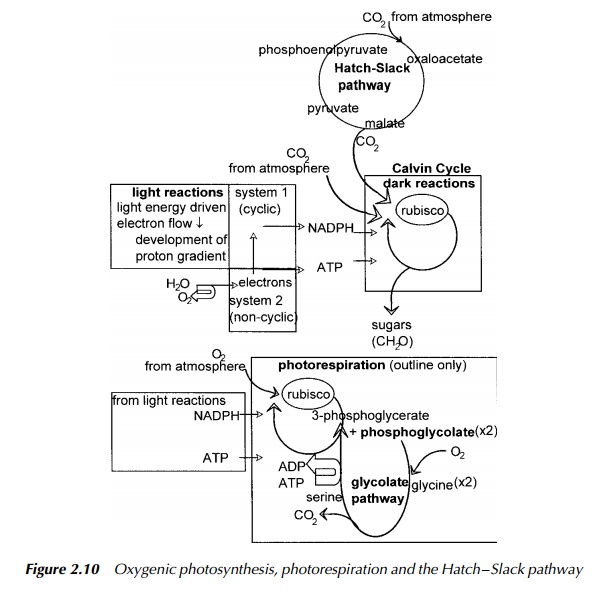
In brief, ribulose diphosphate is carboxylated with carbon dioxide catalysed by the enzyme rubisco to form an unstable six-carbon sugar which is then cleaved to form two molecules of 3-phosphoglycerate, an intermediate of glycolysis. This is not the only route of entry of carbon dioxide into carbohydrate synthesis, the other being the Hatch – Slack pathway. This subject is discussed in more detail later.
Returning to the Calvin cycle, rearrangements of 3-phosphoglycerate produced by rubisco then take place by similar steps to the reversible steps of glycoly-sis and reactions of the pentose phosphate pathway. After completion of three cycles, the net result is the fixation of three molecules of carbon dioxide into a three-carbon sugar, each cycle regenerating a ribulose phosphate molecule. After phosphorylation of the trioses at the expense of ATP, they may enter into glycolysis and be converted into glucose and then to starch to be stored until required. The Calvin cycle is so familiar that it is easy to overlook the fact that not all reducing equivalents are channelled through rubisco to this cycle and carbohydrate synthesis.
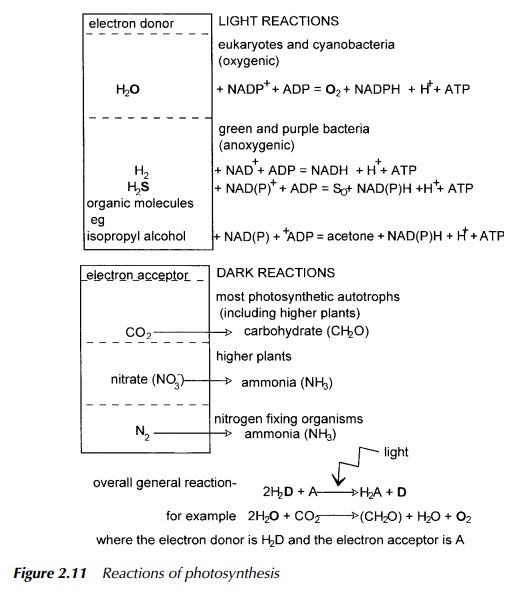
Some organisms may use additional pathways involving other electron accep-tors such as nitrate, nitrogen and hydrogen atoms, the reduction of which obvi-ously does not produce carbohydrate but different essential nutrients which may then also be available to other organisms. These products are summarised in Figure 2.11. For example, when nitrogen or nitrate is the electron donor, the product is ammonia which becomes incorporated by amino transfer into amino acids and thus forms part of the nitrogen cycle. Nitrogen is a particularly note-worthy case in the context of this book as it is the process of nitrogen fixation. This is performed by a number of nitrogen-fixing bacteria some of which are free living in the soil and some form symbiotic relationships with certain leguminous plants, forming root nodules. Nitrogen fixation is, by necessity, an anaerobic pro-cess, and so one essential role for the plant is to provide a suitable oxygen-free environment for these bacteria, the other, to provide energy. Genetic manipula-tion of plants is discussed but it is relevant to point out here that the suggestion is often mooted of introducing the genes responsible for allowing nitrogen fixation to be transferred from the relevant bacteria into suitable plants. The symbiotic relationship between plant and bacteria is very difficult to create artificially and has been a stumbling block in the drive to increase the number of plant species able to host nitrogen fixation. The complicated interaction between plant and bacterium involves intricacies of plant physiology as well as genetic capability provided by the bacterium, and so it is unlikely that a simple trans-fer of nitrogen fixation genes from bacterium to plant will be successful. This, however, remains a research area of major importance.
The issue of nitrogenous material, particularly in respect of sewage and asso-ciated effluents, is of considerable relevance to the environmental application of biotechnology. In addition, there is great potential for phytotechnological intervention to control nitrogen migration, most especially in the light of the burgeoning expansion of nitrate-sensitive areas within the context of agricultural fertiliser usage. Hence, bioengineering of the nitrogen cycle, at least at the local level, provides an important avenue for the control of pollution and the mitigation of possible eutrophication of aquatic environments. The cycle itself, and some of the implications arising, are discussed later.
C3 and C4 plants
Plants for which the reaction catalysed by rubisco is the first point of entry of atmospheric carbon dioxide into carbohydrate metabolism are termed C3 plants due to the product of rubisco being two molecules of 3-phosphoglycerate which contains three carbons. This is the typical route for temperate organisms. An alternative to direct carboxylation for introducing carbon dioxide into the Calvin cycle used by some tropical plants, is the Hatch – Slack pathway illustrated in Figure 2.10. In this case the first step of entry for atmospheric carbon dioxide is by carboxylation of phosphoenolpyruvate by phosphoenolpyruvate carboxylase to produce the four-carbon molecule, oxaloacetate. Hence, plants able to use this pathway are termed C4 plants. The oxaloacetate is part of a cycle which carries the carbon dioxide into the bundle-sheath cells and so away from the surface of the plant, to where the oxygen concentration is lower. Here the carbon dioxide, now being carried as part of malate is transferred to rubisco thus releasing pyruvate which returns to the mesophyll cells at the surface of the plant where it is phosphorylated at the expense of ATP to phosphoenolpyruvate ready to receive the next incoming carbon dioxide molecule from the atmosphere.

The overall effect is to fix atmospheric carbon dioxide, transfer it to a site of lower oxygen concentration compared with the surface of the plant, concentrate it in the form of malate and then transfer the same molecule to rubisco where it enters the Calvin cycle. Although the Hatch – Slack pathway uses energy, and therefore may seem wasteful, it is of great benefit to plants growing in the warmer regions of the globe The reason for this is that the enzyme involved in carbon dioxide fixation in C4 plants namely phosphoenolpyruvate carboxy-lase has a very high affinity for carbon dioxide and does not use oxygen as a substrate, contrasting with rubisco. The result of this competition between oxygen and carbon dioxide for binding to rubisco is the futile process of pho-torespiration, described in the next section. The affinity of carbon dioxide for rubisco falls off with increasing temperature and so in a tropical environment, the efficiency of rubisco to fix carbon dioxide is low. In this situation, the dis-advantage in using energy to operate the Hatch – Slack pathway is more than compensated for by the advantage of being able to fix carbon dioxide efficiently at elevated temperatures. So advantageous is this that much research is being directed to transferring the capability to operate the Hatch – Slack pathway into selected C3 plants.
In the broadest sense of environmental biotechnology, the potential maximisa-tion of solar energy usage, either as a means to the remediation of contamination or to reduce potential pollution by, for example, excessive fertiliser demand, could be of considerable advantage. Hence, appropriately engineered C3 plants in either role offer major advantages in solar efficiency, which, in temperate climes, could provide significant environmental benefits.
Photorespiration
Returning to synthesis of carbohydrate by the Calvin cycle, as mentioned above, the first step is the carboxylation of the five-carbon sugar, ribulose diphosphate catalysed by rubisco. As mentioned in the preceding section, this enzyme may also function as an oxidase indicated by its full name ribulose diphosphate car-boxylase oxidase. When this occurs and oxygen replaces carbon dioxide, the ensuing reaction produces phosphoglycolate in addition to 3-phosphoglycerate. Since, as a result of illumination, oxygen is consumed and carbon dioxide is released during the reactions of the glycolate pathway, this process is termed photorespiration and occurs alongside photosynthesis. The higher the ambient temperature in which the organism is growing, and the higher the oxygen con-centration relative to carbon dioxide, the more pronounced the oxidase activity becomes and consequently the less efficient rubisco is at introducing carbon dioxide into carbohydrate synthesis. The phosphoglycolate formed as a result of oxygen acting as substrate for rubisco, is then dephosphorylated to form gly-colic acid. There follows a series of reactions forming a salvage pathway for the carbons of glycolic acid involving transfer of the carbon skeleton to the per-oxisomes, then to the mitochondria, back to the peroxisomes and finally back to the chloroplast in the form of glycerate which is then phosphorylated at the expense of ATP to re-enter the Calvin cycle as 3-phosphoglycerate. The result of this detour, thanks to the oxidase activity of rubisco is to lose a high energy bond in phosphoglycolate, to consume ATP during phosphorylation to produce 3-phosphoglycerate, consumption of oxygen and release of carbon dioxide. This pathway resulting from the oxidase activity of rubisco, shown in Figure 2.10 is wasteful because it consumes energy obtained by the light reactions with no concomitant fixation of carbon dioxide into carbohydrate. The C3 plants are there-fore operating photosynthesis under suboptimal conditions especially when the oxygen tension is high and carbon dioxide tension is low. Why rubisco has not evolved to lose the oxidase activity is unclear: presumably evolutionary pres-sures of competition have been insufficient to date. For the reasons indicated in the preceding section, C4 plants show little or no photorespiration due to their ability to channel carbon dioxide to rubisco by a method independent of oxygen tension. Therefore they are considerably more efficient than their C3 counterparts and may operate photosynthesis at much lower concentrations of carbon dioxide and higher concentrations of oxygen. It is interesting to contemplate the compet-itive effects of introducing C4 style efficiency into C3 plants, but at the moment this is just speculation.
Balancing the light and the dark reactions in eukaryotes and cyanobacteria
Using the six-carbon sugar, glucose, as an example, synthesis of one molecule requires six carbon dioxide molecules, 12 molecules of water, 12 protons,18 molecules of ATP and 12 molecules of NADPH. Since photophosphorylation is driven by a proton gradient established during electron flow after illumination, there is not a stoichiometric relationship between the number of photons exciting the systems and the amount of ATP produced. However, it is now established that for every eight photons incident on the two photosystems, four for each system, one molecule of oxygen is released, two molecules of NADP+ are reduced to NADPH and approximately three molecules of ATP are synthesised. Since this may leave the dark reactions slightly short of ATP for carbohydrate synthesis, it is postulated that photosystem 2 passes through one extra cycle thus producing additional ATP molecules with no additional NADPH.
Related Topics