Chapter: Introduction to Human Nutrition: Nutrition and Metabolism of Lipids
Reverse cholesterol transport (high-density lipoprotein pathway)
Reverse cholesterol transport
(high-density lipoprotein pathway)
The removal of cholesterol from tissues back to the liver via
HDLs represents the only route of elimina-tion for cholesterol from the body.
This physiological role of HDLs explains, in part, the cardioprotective effects
of these lipoproteins, as indicated by a strong inverse relationship between
serum HDL cholesterol
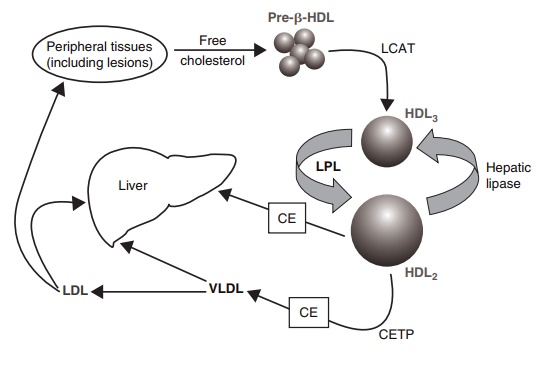
Figure 6.9 Reverse cholesterol trans-port. CE, cholesterol ester;
CETP, choles-terol ester transfer protein; HDL, high-density lipoprotein; LCAT,
lecithin– cholesterol acyltransferase; LDL, low-density lipoprotein; LPL,
lipoprotein lipase; VLDL, very low-density lipoprotein.
The activity of the
HDL pathway is influenced by genetic and dietary factors that can interact to
either increase or reduce the efficiency of cholesterol removal. This, in turn,
may be reflected in changes in the concentra-tion of serum HDLs and their
functional properties.
HDLs are synthesized in the gut and liver, and increase their
particle size in the circulation as a result of the acquisition of cholesterol
from two principal sources: (1) surface material released from TAG-rich
lipoproteins during lipolysis and (2) peripheral tissues. The particles, which
are responsible for removing cholesterol from cells, are very small pre-HDLs
and are disk-shaped particles composed of phospholipid and apoA-I (ApoA-I is
capable of this function on its own). The efflux of free cholesterol from
tissue sites, including deposits of cholesterol in the coronary arteries, is
facilitated by the formation of a free cholesterol gradient from the cell
across the cell membrane to pre-HDLs. The gradient is gener-ated by the
re-esterification of free cholesterol by the enzyme lecithin–cholesterol
acyltransferase (LCAT) and via the migration of these newly formed choles-terol
esters into the hydrophobic core of what becomes mature, spherical HDL. The
newly acquired choles-terol is transported back to the liver either directly by
HDL or indirectly by transfer to apoB-containing lipoproteins VLDL and LDL.
Blood vessels in the liver contain a close relative of LPL, i.e., HL. This
enzyme acts on smaller lipoproteins and especially the surface phospholipid of
HDL, where it effectively punches a hole in the surface coat to facilitate
access to the lipid core and delivery of CE to the hepatocyte (Figure 6.9).
Related Topics