Chapter: Introduction to Human Nutrition: Nutrition and Metabolism of Lipids
Postprandial lipemia
Postprandial lipemia
The turbidity or milkiness of serum or plasma follow-ing the
ingestion of fat marks the arrival of dietary fat now contained in chylomicrons
in the blood. The milky appearance of plasma or serum after the inges-tion of
fat arises from the chylomicrons, which are of a sufficient size physically to
scatter light and create the milky appearance of serum or plasma after a meal.
The size and composition of the chylomicrons pro-duced after a fatty meal are
determined by the fat content of the meal. Hence, the nature of fatty acids in
chylomicron TAG reflects the nature of fatty acid in the meal. Each chylomicron
particle carries a single molecule of apoB-48 which, unlike its other smaller
counterparts A-I and A-IV, remains with the chylomi-cron throughout its life in
the circulation. There is little evidence to suggest that the production of
apoB-48, and thus the number of particles, increases in response to an
increased flux of dietary fat. Instead, the enterocyte incorporates more TAG
into each chy-lomicron and expands the size of each chylomicron to facilitate
the transport of larger amounts of absorbed dietary fat. There is evidence to
suggest that chylomicrons containing lipids enriched with poly-unsaturated
fatty acids (PUFAs) are larger than chylomicrons enriched with saturated fat,
since the former occupy more space when packaged into a lipo-protein. This has
implications for the subsequent metabolism and fate of these lipoproteins in
the cir-culation, since TAGs associated with larger chylomi-crons are known to
be hydrolyzed more rapidly. It is thought that apoB-48 is produced continuously
in the enterocyte-forming pools of apoB-48 in readiness for the sudden
reception of dietary fat and production of chylomicrons. Nevertheless, small
chylomicrons can be detected throughout the postabsorptive phase.
The onset, duration, and magnitude of postpran-dial lipemia can
be monitored in the laboratory after a standard fat-containing meal by making
serial mea-surements of serum TAG or more specifically TAG associated with
TAG-rich lipoproteins over a post-prandial period of up to 8 or 9 hours
(remnants of chylomicrons can be detected 12 hours after a meal).
Alternatively, the levels of apoB-48 or retinyl esters in serum act as useful
markers or tracer molecules for following the metabolism of chylomicrons in the
postprandial period. In normal subjects postpran-dial lipemia peaks between 3
and 4 hours and sub-sides to baseline concentration after 5–6 hours. In some
cases, postprandial TAG (mainly in chylomi-crons) can appear in the blood
within 30 min and peak as early as 1 hour after the ingestion of fat. So rapid
is this rise in TAG that it is believed to represent preformed lipid in the
enterocyte from the previous meal that is shunted into the circulation by the
incom-ing fat load. Note that, in addition to the time taken to emulsify,
hydrolyze, and absorb dietary fat, re-esterification of TAG in the enterocyte
and lipopro-tein assembly alone takes about 15 min, although shunting means
that the first TAG can appear within 30 min, with the first peak after 1 hour.
This shunting phenomenon is particularly noticeable during the day and gives
rise to two or even more peaks, whereas postprandial peaks following an
overnight fast are usually monophasic.
Chylomicrons are not the only TAG-rich lipopro-teins in the
postprandial phase. Chylomicrons clearly contribute significantly to the extent
of postprandial lipemia, and the rate at which the TAGs in these lipo-proteins
are hydrolyzed is known to be a critical deter-minant of the extent and time-course
of postprandial lipemia. The TAGs in circulating chylomicrons are
lipolyzed by a rate-limiting lipase known as lipopro-tein lipase
(LPL). LPL is tethered to the endothelial lining of blood vessels in peripheral
tissues, most notably muscle and adipose tissue, by proteoglycan fibers, and as
such is known as an endothelial lipase. Several molecules of LPL can interact
and lipolyze the TAG from a single chylomicron particle to generate a
chylomicron remnant which is removed by specific cell membrane receptors in the
liver. The situation is com-plicated by the fact that TAG-rich lipoproteins
from the liver, known as very low-density lipoprotein (VLDL), also contribute
to this postprandial lipemia to variable extents in health and disease states.
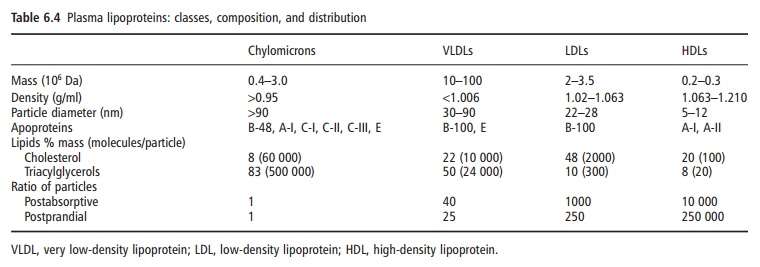
These VLDLs containing endogenously produced TAG are similar in lipid
composition to chylomicrons but con-siderably smaller (Table 6.4). While
chylomicrons carry up to 80% of measurable plasma TAG during the postprandial
period, VLDL particles can carry up to 80% of the measurable protein (mainly as
apo-B), and significantly outnumber chylomicrons at all times. VLDL-TAG are
also metabolized by LPL, which creates competition for the clearance of
endogenously and exogenously derived TAG carried by VLDLs and chy-lomicrons
respectively.
Related Topics