Chapter: Introduction to Human Nutrition: Nutrition and Metabolism of Lipids
Re-esterification of triacylglycerols in the enterocyte
Re-esterification of triacylglycerols
in the enterocyte
Once LCFAs have entered the cell they are activated by acyl-CoA
and are re-esterified with glycerol back into TAG and phospholipids by two
distinct bio-chemical pathways, the 2-MAG and glycerol-3-phosphate (G-3-P)
pathways. The difference between these two pathways lies in:
● their substrates of activation
● the former using 2-MAG and the latter α-glycero-3-phosphate
● their location within different cellular organelles: the 2-MAGs
reside in the smooth endoplasmic reticulum and the G-3-P in the rough
endoplasmic reticulum
● the periods during which they are most active.
The 2-MAG pathway is quantitatively of greater importance in the
enterocyte of the intestine and thus predominates in the postprandial period,
whereas the G-3-P pathway is more active in the postabsorptive phase in tissues
such as liver, muscle, and adipose tissue. Following the absorption of a fatty
meal and uptake of 2-MAG into the enterocyte, up to 90% of these molecules are
rapidly acylated back to 1,2-dia-cylglycerol and finally TAGs by the sequential
actions of three enzymes: CoA ligase, monoglycerol acyl-transferase, and
diacylglycerol acyltransferase. In a similar fashion, lysophosphatidylcholine,
produced by the action of pancreatic phospholipase ‘A’ on dietary
phospholipids, is absorbed by the enterocyte and re-esterified back to
phosphatidylcholine in the enterocyte by direct acetylation (Figure 6.7). The
bulk of free cholesterol absorbed from the intestinal lumen is also
re-esterified in the enterocyte by the enzyme ACAT.
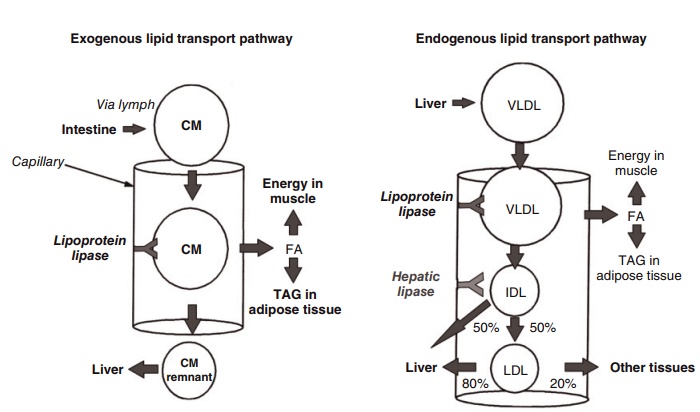
Figure 6.7 Re-esterification of triacylglycerides in enterocytes. CM, chylomicron; FA, fatty acid; IDL, intermediate-density lipoprotein; LDL, low-density lipoprotein; TAG, triacylycerol; VLDL, very low-density lipoprotein. (Reproduced from Mangiapane EH, Salter AM, eds. Diet, Lipoproteins and Coronary Heart Disease. A Biochemical Perspective. Nottingham University Press, Nottingham, 1999, with permission of Nottingham University Press.)
Related Topics