Chapter: Genetics and Molecular Biology: Repression and the lac Operon
Repressor Binds to DNA: The Operator is DNA
Repressor Binds to DNA: The Operator is DNA
We now know that repressor acts by
binding to the operator and preventing RNA polym-erase from binding to the lac promoter. The first experiments to
show that repressor binds to lac DNA
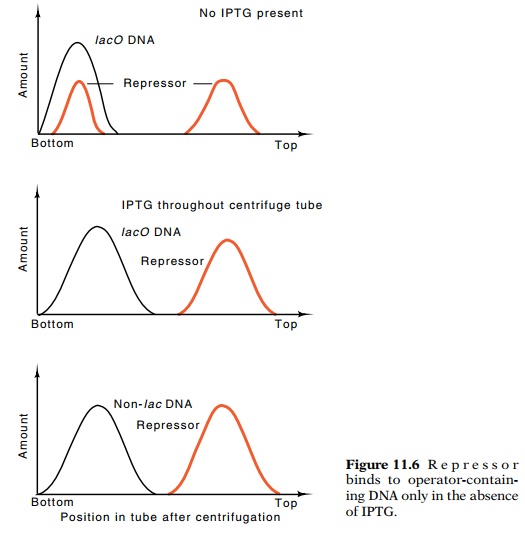
used ultracentrifugation of radioactive repressor and the DNA from λlac phage.
The DNA sediments at 40S, whereas repressor sediments at 7S. If repressor bound
so tightly to DNA that it did not come off during the centrifugation, it would
sediment at about 40S. Indeed, DNA containing lac carried lac repressor
along with it down the centrifuge tube, but only if inducers of the lac operon were not present (Fig. 11.6).
These are the properties expected of repressor if it were to regulate by
binding to DNA to prevent transcription.
A
considerably easier assay of DNA binding by lac
repressor was developed later by Riggs and Bourgeois. Pure lambda DNA passes
through cellulose nitrate filters, but repressor, like many other proteins,
binds to
the filters. It is bound by molecular interactions and not by filtration. Most
surprisingly, if repressor is bound to DNA, the DNA molecule will not pass
through the filter either. Thus if the DNA is made radioactive, the retention
of lac DNA on the filter can easily
be detected, providing a simple assay for repressor binding to operator (Fig.
11.7).
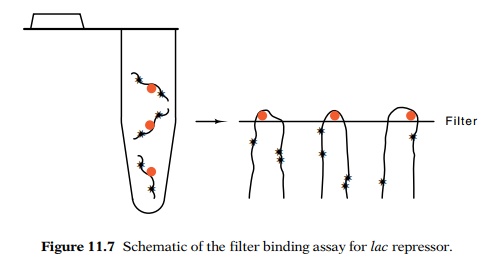
Figure 11.7 Schematic of the filter binding assay forlacrepressor.
The
filter-binding assay is very sensitive because the DNA can be made highly
radioactive with 32PO4 The long DNA molecule contains
many phosphate groups, and the half-life of 32P is short. This assay facilitated measurement of the
rates of binding and dissociation as well as a determination of the equilibrium
constant for binding.
For example, the rate of lac repressor dissociation from operator is easily measured by mixing repressor with radioactive DNA so that the majority of repressor is bound to operator. Then the solution is diluted and a large excess of nonradioactive lac DNA is added. Any repressor that subsequently dissociates from operator either remains free in solution or has a much greater chance of binding to nonradioactive DNA. At intervals after the dilution, the mixture is filtered to determine the fraction of repressor still bound to the radioactive lac operator-con-taining DNA. The kinetics in the reduction in the amount of radioactive DNA bound to the filters with increasing time give the dissociation rate of repressor from operator.
Related Topics