Chapter: Genetics and Molecular Biology: Repression and the lac Operon
Proving lac Repressor is a Protein
Proving lac
Repressor is a Protein
The phenomenon of repression in the lac operon was characterized by genetic experiments. These showed that a product of the lacI gene diffused through the cell and shut off expression of the lac genes. Basically these were complementation experiments that showed that lacI acted in trans to repress. A LacI-LacZ+strain constitutively ex-presses β-galactosidase in the absence or presence of inducers. The lacZ gene in such a strain is repressed if an episome is introduced which carries a good lacI gene but is deleted of the lacZ gene. The genetic structure of the resulting strain is denoted F’lacI+∆lacZ/lacI-lacZ+. That is, LacI protein acts in trans to repress the chromosomal lacZ gene.
Once it
was established that the LacI product repressed expression, research on the lac operon shifted to learning what
repressor was; whether it was RNA or protein, and how it acted to block
expression of the lac enzymes. Since
the target of the repressor was a sequence that could be genetically mapped,
two likely possibilities existed. The opera-tor could be a region on the DNA to
which repressor bound to reduce the intracellular levels of functional lac messenger. Alternatively, re-pressor
recognized and bound to a region on the lac
messenger and reduced translation efficiency. Either of these possibilities
required recognition of a specific nucleotide sequence by the lacI gene product. To many
investigators, such an ability seemed most reasonable for an RNA molecule.
Indeed, early results obtained with RNA and protein synthesis inhibitors led to
the conclusion that it was not protein. Gilbert and Müller-Hill, however,
reasoned that an RNA molecule was unlikely to possess all the regulatory
properties required of the lacI
product. Therefore Müller-Hill designed a simple genetic experiment that
indi-cated that repressor contained at least some protein.
His proof
was that nonsense mutations could be isolated in the lacI gene. A nonsense mutation can only be isolated in a gene that
encodes a protein. The starting point was a strain containing amber mutations
in both the gal and his operons. This strain was then made
LacI- by selection of constitutives. Most of these mutants were
missense, but a few were nonsense I- mutants. Each of the
constitutives was then reverted simultaneously to Gal+ and His+.
The only way a cell could perform this double reversion was to become nonsense
suppressing, Su+, and simultaneously suppress both nonsense
mutations. These nonsense-suppressing revertants were then tested for lac constitutivity,
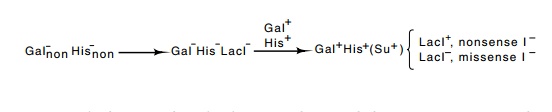
and a few
were found to have simultaneously become I+. Consequently, these
were nonsense I mutations that were now being suppressed and were forming
functional repressor that turned off expression of the lac operon in the absence of inducers. This proves that the I gene
product contains protein, but it does not prove that the repressor is entirely
protein. The repressor had to be purified before it could be shown to be
entirely protein.
Related Topics