Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Viral Multiplication
Release - Viral Multiplication
RELEASE
Bacteriophages
Most bacteriophages escape from the infected cell by coding for one or more enzymes synthesized late in the latent phase that causes the lysis of the cell. The enzymes are either lysozymes or peptidases that weaken the cell wall by cleaving specific bonds in the peptidoglycan layer. The damaged cells burst as a result of osmotic pressure.
Animal Viruses
CELL DEATH
Nearly all productively infected cells die (see below for exceptions), presumably be-cause the viral genetic program is dominant and precludes the continuation of normal cell functions required for survival. In many cases, direct viral interference with nor-mal cellular metabolic processes leads to cell death. For example, picornaviruses shut off host protein synthesis soon after infection, and many DNA animal viruses interfere with normal cell cycle controls. In many cases, the end result of such insults is a trig-gering of a cellular stress response called programmed cell death or apoptosis.Some viruses are known to code for proteins that block or delay apoptosis, probably to stave off cell death until the virus replication cycle has been completed. Ultimately, the cell lysis that accompanies cell death is responsible for the release of naked capsid viruses into the environment.
BUDDING
With the exception of the poxviruses, all enveloped animal viruses acquire their membrane by budding either through the plasma membrane or, in the case of herpesviruses, through the membrane of an exocytic vesicle. Thus, for these viruses, release from the cell is coupled to the final stage of virion assembly. How the herpesviruses ultimately escape from the cell when the membrane of the exocytic vesicle fuses with the plasma membrane. The poxviruses appear to program the synthesis of their own outer membrane. How the poxvirus envelope is assembled on the nucleocapsid is not known.
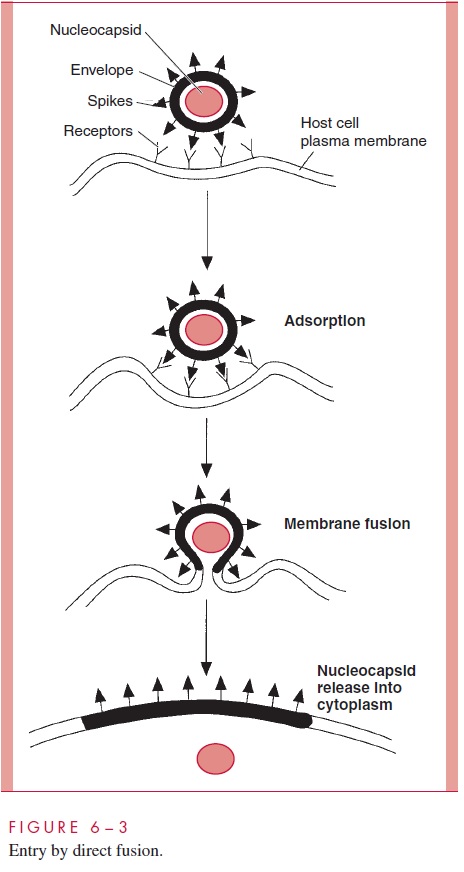
The membrane changes that accompany budding appear to be just the reverse of the entry process described before for those viruses that enter by direct fusion (compare Fig 6 – 3 and Fig 6 – 11). The region of the cellular membrane where budding is to occur
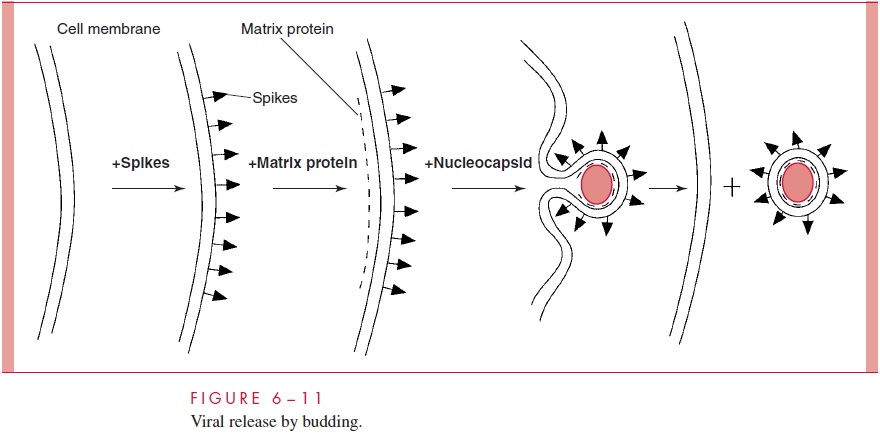
These proteins are synthesized by the pathway that normally delivers cellular membrane proteins to the surface of the cell by way of the Golgi apparatus. At the site of the glycoprotein cluster, the inside of the mem- brane becomes coated with a virion structural protein called the matrix or M protein. The accumulation of the matrix protein at the proper location is probably facilitated by the presence of a binding site for the matrix protein on the cytoplasmic side of the trans- membrane glycoprotein spike. The matrix protein attracts the completed nucleocapsid that triggers the envelopment process leading to the release of the completed particle to the outside (see Fig 6 – 11).
For viruses that bud, it is important to note that the plasma membrane of the infected cell contains virus-specific glycoproteins that represent foreign antigens. This means that infected cells become targets for the immune system. In fact, cytotoxic T lymphocytes that recognize these antigens can be a significant factor in combating a virus infection. The process of viral budding usually does not lead directly to cell death because the plasma membrane can be repaired following budding. It is likely that cell death for most enveloped viruses, as for naked capsid viruses, is related to the loss of normal cel-lular functions required for survival or as a result of apoptosis. Unlike most retroviruses that do not kill the host cell, HIV-1 is cytotoxic. Although the mechanism of HIV-1 cell killing is not entirely understood, factors such as the accumulation of viral DNA in the cytoplasm, the toxic effects of certain viral proteins, alterations in plasma membrane permeability, and cell – cell fusion, are believed to contribute to the cytotoxic potential of the virus.
CELL SURVIVAL
For retroviruses (except HIV-1 and other lentiviruses) and the filamentous bacteriophages, virus reproduction and cell survival are compatible. Retroviruses convert their RNA genome into double-stranded DNA, which integrates into a host cell chromosome and is transcribed just like any other cellular gene . Thus, the impact on cellular metabolism is minimal. Moreover, the virus buds through the plasma membrane without any permanent damage to the cell.
Because the filamentous phages are naked capsid viruses, cell survival is even more remarkable. In this case, the helical capsid is assembled onto the condensed single-stranded DNA genome as the structure is being extruded through both the membrane and the cell wall of the bacterium. How the cell escapes permanent damage in this case is un-known. As with the retroviruses, the infected cell continues to produce virus indefinitely.
Related Topics