Chapter: Pharmaceutical Drug Analysis: Errors In Pharmaceutical Analysis and Statistical Validation
Recommendations for Criteria of Rejecting an Observation
RECOMMENDATIONS FOR CRITERIA OF REJECTING AN OBSERVATION
An analyst, while carrying out a series of measurements,
invariably comes across with ONE specific result in a set of replicates that
obviously appears to be quite ‘out of place’ with the others, and at this
juncture he should take an appropriate decision whether to discard (or expunge)
this result from any further consideration. Thus, two situations often arise, namely :
(i) Number of
replicates being small, and
(ii) Number of
replicates being large.
A. Number of Replicates being Small
An analyst in the true sense encounters a serious problem
when the number of replicates at his disposal is SMALL. Firstly, the divergent
result shows a distinct and significant effect upon the mean value ( x’) ; and secondly, the prevailing
scanty available data does not permit getting at the real statistical analysis
of the status of the suspected result.
B. Number of Replicates being Large
In this instance, the analyst has the privilege of
rejecting one value (i.e., the
‘out-of place’ value) as it is not an important one by virtue of the following
two main reasons :
Firstly, a single value shall exert merely a small effect
upon the mean value ( x ) ; and
secondly, the treatment of data with the real statistical analysis would
certainly reveal vividly the probability that the suspected ‘out of place’
result is a bonafide member of the same population as the others.
![]()
Blaedel et al.*
(1951), Wilson** (1952) and Laitinen*** (1960) have put forward more broadly
accepted and recommended criteria of rejecting an observation.
1. Rules Based on the Average Deviation
Both ‘2.5d’ and
‘4d’ rules are quite familiar to
analysts. They may be applied in a sequential manner as follows :
(i) Calculate
the mean ( x’ ) and average deviation
( d’ ) of the ‘good’ results,
![]()
![]()
(ii) Determine
the deviation of the ‘suspected’ result from the mean of the ‘good’ results,
(iii) In case,
the deviation of the suspected result was found to be either 2.5 times the
average deviation of the good results (i.e.,
‘2.5d’ rule) or 4 times the average
deviation of the good results (i.e.‘4d’ rule) the suspected result was rejected out right ; otherwise the
result was duly retained.
Note : The ‘limit for rejection’
seems to be too low for both the said rules.
2. Rules Based on the Range
The Q test, suggested by Dean and Dixon**** (1951) is
statistically correct and valid, and it may be applied easily as stated below :
(i) Calculate
the range of the results,
(ii) Determine
the difference between the suspected result and its closest neighbour,
(iii) Divide
the difference obtained in (ii) above
by the range from (i) to arrive at
the rejection Quotient Q,
(iv) Finally,
consult a table of Q-values. In case, the computed value of Q is found to be
greater than the value given in the table, the result in question can be
rejected outright with 90% confidence that it was perhaps subject to some
factor or the other which never affected the other results.
Table 3.2, records some of the Q-values as given below :
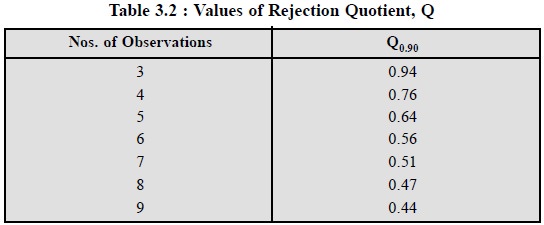
Example : Five determinations of the
ampicillin content in capsules of a marketed product gave the following
results : 0.248, 0.245, 0.265, 0.249 and 0.250 mg per capsule. Apply the Q-test
to find out if the 0.265 value can be rejected.
The value of Q is :
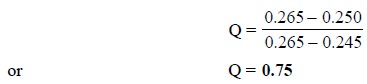
The value in Table 3.2, at n = 5 is Q = 0.64. Because, the determined value 0.75 > 0.64,
according to ‘rule based on the range’ the result i.e., 0.265 can be rejected.
Note : The Q-test administers
excellent justification for the outright rejection of abnormally erroneous
values ; however, it fails to eliminate the problem with less deviant
suspicious values.
Related Topics