Chapter: Pharmaceutical Biotechnology: Fundamentals and Applications : Recombinant Coagulation Factors and Thrombolytic Agents
Recombinant Coagulation Factors and Thrombolytic Agents
INTRODUCTION
Coagulation and fibrinogenolysis exist in a mutually compensatory or balanced state. Endogenous regulatory mechanisms ensure that the processes of hemostasis and blood coagulation at a site of injury, and the subsequent fibrinolysis of the blood clot, are localized and well controlled. This ensures a rapid and efficient hemostatic response at a site of injury while avoiding thrombogenic events at sites distant from the site of injury, or the hemostatic response persisting beyond its physiologic need. This chapter will focus on recombinant products that are available to facilitate coagulation and for thrombolysis.
Blood coagulation has been divided into the extrinsic (tissue factor dependent) and intrinsic path-ways that converge to the common pathway, leading to the generation of thrombin (Fig. 1). More recently, a revised cell-based model of coagulation has been proposed (Hoffman and Monroe, 2005). This cell-based model emphasizes the interaction of clotting factors with cell surfaces, and appears to explain someof the unresolved issues with the cascade model (Hoffman, 2003).
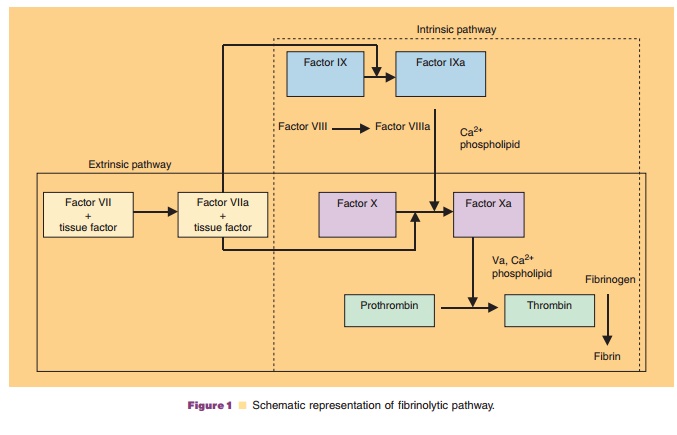
Normally hemostasis is a highly efficient and tightly regulated process to ensure that it occurs quickly and is localized. Abnormalities that result in a delay in blood coagulation are associated with a bleeding tendency termed hemophilia. Hemophilia is an X chromosome-linked recessive disorder. The incidence of hemophilia is estimated at 5 to 6 people per 100,000 males. Hemophilia A (classical hemophi-lia) patients have decreased, defective, or absent production of factor VIII, whereas patients with hemophilia B lack factor IX. Factor XI deficiency (originally termed hemophilia C) is less common and in most cases is a mild bleeding disorder. The availability of recombinant coagulation factors has been a major advance in the area of hemophilia, providing the promise of unlimited supply, ease of use, improved safety, and reducing the risk of infections transmitted by transfusion.
Related Topics